‘To be or not to be’ a standardized botanical powder

Dr Raina Raj, Head of Marketing, Natural Remedies Pvt. Ltd.
 The demand for organic and natural foods has grown over the past decade. The desire for better overall health among consumers and the awareness of the harmful effects of synthetic ingredients has fuelled an organic revolution. This is evident from the global market estimations; “the organic food market generated $162,036.7 million in 2019 and is predicted to make revenue of $416,049.7 million by 2027.” To be a front player in the future of the organic revolution, consistency in quality and biological results shall be essential. Keeping this in mind, we have designed a series of articles describing the importance of the standardization of botanical powders. Standardization here refers to neutralizing the natural variability to deliver consistency and uniformity in the quality assessment parameters. According to the National Institutes of Health (NIH), standardization is a process that manufacturers use to ensure batch-to-batch consistency of their products. Standardization involves identifying specific phytochemicals (also known as markers) that can be used to manufacture a consistent product. In our earlier article, we discussed what and how standardization is done in standardized botanical powders (SBPs).
The demand for organic and natural foods has grown over the past decade. The desire for better overall health among consumers and the awareness of the harmful effects of synthetic ingredients has fuelled an organic revolution. This is evident from the global market estimations; “the organic food market generated $162,036.7 million in 2019 and is predicted to make revenue of $416,049.7 million by 2027.” To be a front player in the future of the organic revolution, consistency in quality and biological results shall be essential. Keeping this in mind, we have designed a series of articles describing the importance of the standardization of botanical powders. Standardization here refers to neutralizing the natural variability to deliver consistency and uniformity in the quality assessment parameters. According to the National Institutes of Health (NIH), standardization is a process that manufacturers use to ensure batch-to-batch consistency of their products. Standardization involves identifying specific phytochemicals (also known as markers) that can be used to manufacture a consistent product. In our earlier article, we discussed what and how standardization is done in standardized botanical powders (SBPs).
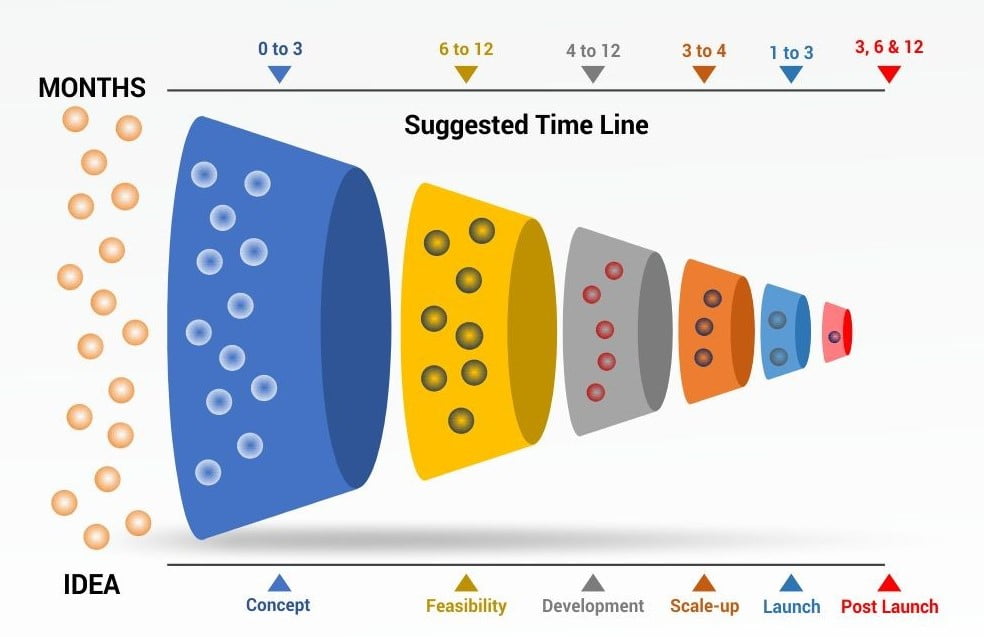
In the current article, we shall discuss how an SBP comes into being. What are the process and aggressive scrutinization that occurs at each step of its formulation from when it is an idea to its launch into the market? Shakespeare’s hamlet phrase “to be or not be” fits in aptly here. The well-known stage-gate process for innovative products can be used as a basis with slight modifications (Figure 1). At each stage a committee would review the process and approve or disapprove the SBP from entering the next stage.
Ideation stage: In the ideation stage, an idea of a product is proposed to address a pain point or an issue, in the poultry industry. Either to bring in a new supplement or to find a natural, and safe alternative to an existing synthetic supplement in the market.
Concept stage: A clear customer value proposition for the idea is made through a business case. What would the impact of this SBP be in the market? A library of plants is created based on availability, biodiversity, patentability, and cost. If the SBP passes this stage, it goes to the feasibility stage.
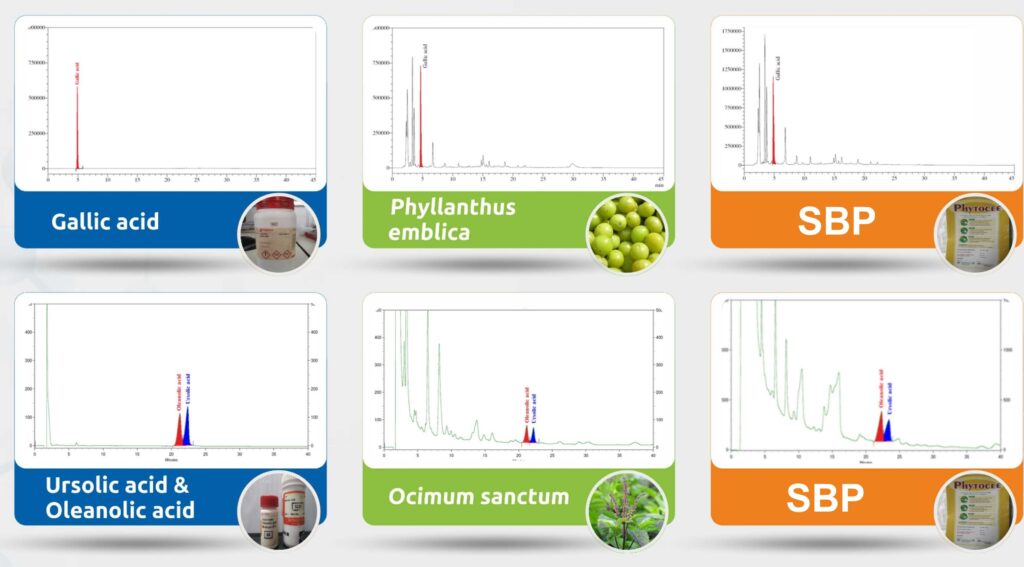
Feasibility stage: The potential plants that would address the issue are screened. Medicinal plants are screened with bioassays in vitro or in vivo. Traditional and modern scientific literature is curated for botanical constituents that can be used to address and solve the issue. The phytochemistry laboratory develops commercially viable processes for the optimum extraction of medicinal plants, where active constituents, otherwise known as markers, are already known. The phytochemistry lab is where the creation of a SBP starts, with the preparation of different formulations. The SBPs are prepared through scientific evaluation of the phytochemicals required for the formulation and the right selection of the raw materials. The phytochemistry lab also establishes the essential reference marker or the phytochemical that would be necessary to be standardized in each batch of SBPs (Figure 2).
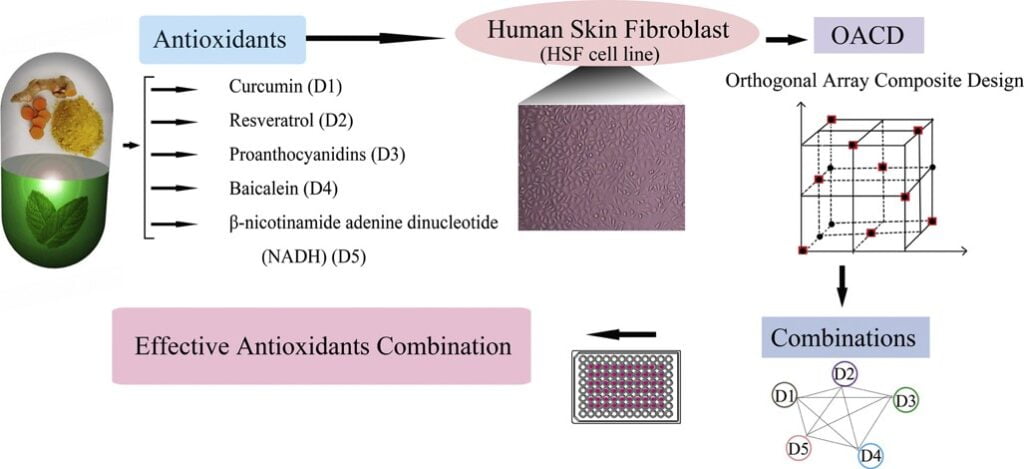
Developmental stage: Leads generated in the feasibility stage are examined and combined to form formulation prototypes. This is performed with computational models such as orthogonal array composite design (OACD) (Figure 3), which helps look for interactions between the different components of the tentative formulations. These results simulate the interaction between the components and give a scientific insight regarding the interaction between the constituents. They could either be synergistic, additive or antagonistic. Additivity means that the activity of the ingredients is adding up, synergy means the activity of the components when combined is enhanced, and antagonism means that the ingredients would nullify the effects of each other. Only those combinations that show synergistic predictions in a formulation are prioritized and selected to be taken further.
A series of biological studies are conducted to find the best herb(s) for the desired biological activity. The SBP formulation is then tested in a small group of the target species in a controlled environment. It is assessed for safety in the animals. Only if these studies show promising results they move to the scale-up stage.
Scale-up stage: The challenges during manufacturing are addressed in this stage, where the product is scaled in quantity from kilograms to tons. Then, the beta-testing phase begins, where a larger number of target species is tested. Farm trials in field conditions are conducted in different parts of the country and the world. If the results are encouraging at the scale-up stage, the product is launched to a segment of the market.
Launch stage: In the launch stage the SBP is evaluated for its safety, effectiveness and checked if it meets all the end attributes that were desired in the conceptual stage. Later on, it is taken to full-scale marketing.
Post-marketing surveillance: Test to examine how the SBP fairs. Is the SBP efficient in showing the desired biological effects? Its mechanisms of action at the phenotypic, molecular, and genome level are evaluated.
Only when an SBP can pass through each stage with satisfactory performance, the process is furthered to the next stage, if not the journey of the SBP is halted; to iterate its formulations and the journey begins from the start with new constituents. In our next article, we shall elaborate on when the standardization process begins “Standardization of SBP starts at the grassroots”