MECHANISM OF ANIBIOTIC RESISTANCE
MECHANISM OF ANIBIOTIC RESISTANCE
Prof.Dr. R.N.Sreenivas Gowda
Former and Founding VC, KVAFSU, Bidar, Former Director, IAH&VB, Bangalore, Former Prof and University Head, Dpt. Of Pathology, Veterinary College, UAS, Bangalore
Introduction
Demand for animal protein for human consumption is rising globally at an unprecedented rate and has significant public health implications. The global expansion of intensive farming has led to an increase in antimicrobial use to contain bacterial diseases The modern animal production practices are also associated with regular use of antimicrobials to prevent diseases and promote growth potentially increasing selection pressure on bacteria to become resistant.
Antibiotics have been started using around 1928 when penicillin was first discovered by Alexander Fleming. In the 1980s, antibiotics that were determined medically important for treatment of animals could be approved under veterinary guidance. In 1996, the National Antimicrobial Resistance Monitoring System (NARMS) was established. Starting in 2010, publications regarding antimicrobial drugs in food became an annual report. Starting in 2012, there was publicly solicited input on how data is to be collected and reported for matters relating to the use of antimicrobials for food-producing animals.
An analysis of Indian Pharmaceutical market has found that 70.4% of the fixed antibiotic formulations present in the market in 2020 were either unapproved or banned. Despite regulatory initiatives in2007and 2013 many fixed dose combinations are available in the market. The use of such drugs results in antibiotic resistance making infections harder to treat .
It is estimated that the global consumption of veterinary antimicrobials is projected an increase of 11.5% by 2030 to the extent of 104,079 tonnes. It has also been estimated that there will be an increase of 15% for AMR in humans between 2015 and 2030.
Key Factors presented by WHO 2020 on antibiotics are:
- Antibiotic resistance is one of the biggest threats to global health, food security, and development today.
- Antibiotic resistance can affect anyone, of any age, in any country.
- Antibiotic resistance occurs naturally, but misuse of antibiotics in humans and animals is accelerating the process.
- A growing number of infections – such as pneumonia, tuberculosis, gonorrhea, and salmonellosis – are becoming harder to treat as the antibiotics used to treat them become less effective.
- Antibiotic resistance leads to longer hospital stays, higher medical costs and increased mortality.
Based on the above factors this paper provides the insight mechanism of antimicrobial resistance in animals and human medicine, in short it is called as AMR .
What are antibiotics? The term antibiotic as a chemical compound generated from microorganisms that inhibits or destroys other microbes. In other words, antibiotics are chemical substances produced by a living organisms used to prevent and treat bacterial infections in humans and animals either by killing them or making it difficult for their growth and multiplication.
An estimated 131,109 tons of all antimicrobials were used in food animals in 2013 and the figure is projected to rise to 200,235 tons by 2030. The AMR is a global threat that causes 700,000 deaths annually. The burgeoning AMR is estimated to account for 10 million deaths and US$ 100 trillion economic losses every year by 2050 if no urgent actions are taken. The overall global animal consumption of antibiotics attributed to the pig population followed by cattle and poultry (Fig1).
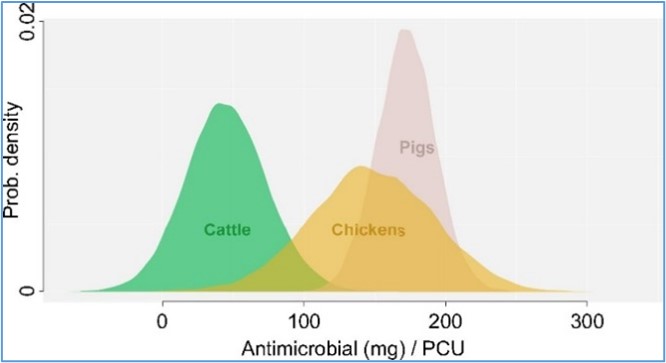
(Source: Thomas P. Van Boeckel et.al.2015 Global trends in antimicrobial use in food animals 112 (18) 5649-5654 https://doi.org/10.1073/pnas.1503141112)
In 2017, China was the largest consumer of veterinary antimicrobials, accounting for 45% of global use, and it is projected to remain the largest consumer in 2030 (43%). The top 10 veterinary antimicrobial consumers in 2017 were: China (45%), Brazil (7.9%), the United States (7.0%), Thailand (4.2%), India (2.2%), Iran (1.9%), Spain (1.9%), Russia (1.8%), Mexico (1.7%), and Argentina (1.5%) (fig.2).
Source: KatieTiseo et,al. PubMed Central (National library of Medicine)
Why antibiotics are used in animal feed?
Antibiotics have been used in animal feed for more than 50 years ever since the discovery not only as an anti-microbial agent, but also as a growth-promoter to improve their performance. Antibiotics are used regularly in animal feed at a rate of 50 to 200 grams per ton for improved performance of animals. The reasons include a more efficient conversion of feed to animal products, an increased growth rate and a lower morbidity/mortality rate in general. The levels are also increased in times of stress and disease. This increased amount is often decreased when the threat of a disease is subsided.
Most commonly used antibiotics in livestock and poultry feed are: Chlortetracycline, Procaine penicillin, Oxytetracycline, Tylosin, Bacitracin, Neomycin sulfate, Streptomycin, Erythromycin, Lincomycin, Oleandomycin, Virginamycin, and Bambermycins.
In addition to these antibiotics, which are of microbial origin, there are other chemically synthesized antimicrobial agents that are also sometimes used in animal feeds. These include three major classes of compounds: Arsenical, Nitro-furan, and Sulfa compounds. Arsenical compounds include arsanilic acid, 3-nitro-4-hydroxy phenylarsonic acid, and sodium arsanilate; nitro-furan compounds include furazolidone and nitro-furazone; sulfamethazine, sulfathiazole, and sulfaquinoxaline. Other chemicals are also used as antiprotozoal agents to prevent coccidiosis and histomoniasis in chickens and turkeys (Table.1,2and 3)
Table 1. Types of antibiotics used in animal farming with their occurrence and behaviour in the environment (based on: Almeida et al. (2014), Sanseverino et al. (2018), Rodriguez-Mozaz et al. (2020) and Mathis et al. (2014)).
| Type of antibiotic | Examples of use in animal breeding | Occurrence in environment veterinary medicine or both human and veterinary areas [μg L−1] or [μg kg−1] | Comments on aquatic environment behaviour |
| Aminoglycosides | Drinking water (e.g., apramycin⁎, streptomycin⁎, spectinomycin⁎) Feed (e.g., neomycin⁎) Injection use |
Not found | Streptomycin: very water soluble, adsorbs strongly onto negatively charged clay soilse Spectinomycin: in storage lagoon concentration decrease (79 %) in the first weekf Neomycin: not expected to adsorb to suspended solids and sediment |
| Amphenicols | Feed (e.g. florfenicol⁎) Injection use |
Florofenicol: Surface water = 0.007–0.93a Drinking water = 0.018–0.211a |
Slow degradation in water environment (>28 days) |
| Cephalosporins | Intramuscular route Injection use |
Cephalexin: Wastewater effluent = 0.3 |
Abiotic processes play an important role in their elimination in surface water Higher degradation at higher pH values |
| Macrolides/lactone polyketides | Drinking water, feed (e.g. tylosin⁎, tilmicosin⁎, spiramycin, erythromycin) Injection use |
Erythromycin: Wastewater = n.d.–7.54 Surface water = 0.0017–0.3 Spiramycin: Wastewater = 0.45–0.60 Surface water = 0.74–3.0 Tilmicosin Wastewater = 0.0035–0.093 Surface water = 0.0025–0.82 Tylosin Surface water = 0.028 |
In soil half-live time 20 daysh, possible migration from soil to groundwateri Amount of spiramycin, dehydro-erythromycin and roxithromycin decreases during treatment process (binding to sludge with later release) |
| Lincosamides | Drinking water, feed (e.g. lincomycin⁎, clindamycin) Injection use |
Lincomycin Wastewater = 0.0061–0.32 Surface water = 0.25 Clindamycin Wastewater = 0.28 |
Half-life time of 30 h, biodegradation is important degradation pathwayj Fast degradation clindamycin in the sediment |
| Penicillins | Drinking water (e.g. amoxicillin, ampicillin, cloxacillin, phenoxy-methylpenicillin⁎) Injection use |
Amoxicillin Wastewater = n.d.–0.04 Surface water = n.d.–5.7 Ampicillin wastewater = 0.25 |
Easily degrading and occasionally detected in environmental samples |
| Quinolones/fluoroquinolones | Drinking water (e.g. ciprofloxacin, enrofloxacin⁎, flumequine⁎, oxolinic acid⁎) Injection or oral use |
Ciprofloxacin Wastewater = n.d.–1.44b Enrofloxacin Wastewater = n.d.–0.18b Surface water = 0.1–0.26 Danofloxacin Surface water = 0.54 Flumequine Wastewater = 0.025 Surface water = 0.01 Oxolinic acid Wastewater = n.d.–0.20b |
Exhibit large chemical stability, bind strongly to sludge Half-life time of 4.8–121 days in seawaterk, l Half-life time of 60–150 days sedimentl, m |
| Sulfonamides/dihydrofolate reductase inhibitors | Drinking water, feed (e.g. sulfadiazine⁎, sulfadoxine, sulfapyridine⁎, trimethoprim) Injection or oral use |
Sulfadiazine Wastewater = 0.00003–0.21 Surface water = 0.004–0.24 Sulfapyridine Wastewater = n.d.–0.33b Trimethoprim Wastewater = 0.00007–4.0 Surface water = 0.00032–0.25 Ground water = 0.0014 Sediment/sludge = 0.0016 |
Sulfonamides (polar and hydrophilic) being transferred into the aquatic environment and they are considered to be the most mobile of antibiotics They are classified as stable substances (app. degradation half-life >1 year) |
| Tetracyclines | Drinking water, feed (e.g. doxycycline⁎, oxytetracycline⁎, tetracycline⁎) Injection or oral use |
Doxycycline Wastewater = n.d.–0.0067 Surface water = 0.19 Oxytetracycline Wastewater = n.d.–0.0013 Surface water = 0.0045–0.041 Ground water = n.d. Sediment/sludge = n.d. Tetracycline Wastewater = n.d.–0.61b Surface water = 0.14 Sediment/sludge = 0.0065 |
Tetracyclines like penicillins, readily precipitate as complex forms with cations (Ca, Mg) and they were accumulated in sludge or sediment |
| Pleuromutilins | Drinking water, feed (e.g. tiamulin⁎, valnemulin) Injection use |
Tiamulin Drinking water = 0.055c |
Half-life time of 10 weeks (lake water) In water solution degraded up to 40 % in 27 days |
| Ionophores | Feed (e.g. monensin⁎, salinomycin⁎, lasalocid⁎) | Monensin Surface water = 0.03d Salinomycin Surface water = 0.04d |
Monensin: half-life in water column ~20 ± 5 days Salinomycin: low solubility in water, high sorption affinity to soil, half-life ~5 days in stored manurep Lasalocid: low solubility in water, high sorption affinity to soil |
Table2. Major Antibiotics Used and their functions in Chickens
| Compound | Growth and Feed Efficiency | Action on Various Infections |
| Bambermycin | yes | no |
| Bacitracin | yes | yes |
| Chlortetracycline | yes | yes |
| Erythromycin | no | yes |
| Gentamycin | no | yes |
| Neomycin | no | yes |
| Novobiocin | no | yes |
| Oleandomycin | yes | yes |
| Oxytetracyclin | no | yes |
| Penicillin | yes | yes |
| Roxarsone | yes | yes |
| Spectinomycin | yes | yes |
| Streptomycin | no | yes |
| Tetracycline | no | yes |
| Tylosin | yes | yes |
| Virginiamycin | yes | yes |
| Fluoroquinolones | no | yes |
(Source: Compiled from FDA Approved Animal Drug List (Green Book), 1998a, and Feed Additive Compendium, 1997).
Table 3. Coccidiostats Used in Broilers (B), Turkeys (T), and Layers (L)
| Sulfonamides | Ionophores | Others |
| Sulfachloropyrazine (B) | Lasalocid (B) | Amprolium (B, T) |
| Sulfamethazine (B, T) | Maduramycin (B) | Arsanilate (B, T, L) |
| Sulfadimethoxine (B, T) | Monensin (B, T) | Buquinolate (B, L) |
| Sulfamyxin (B, T) | Narasin (B, T) | Clopindol (B, T) |
| Sulfanitran (B) | Salinomycin (B, T) | Dequinate (B) |
| Sulfaquinoxaline (B, T) | Nequinate (B) | |
| Nicarbazin (B) | ||
| Robenidine (B) | ||
| Zoalene (B, T) |
(Source: Compiled from FDA Approved Animal Drug List (Green Book), 1998a, and Feed Additive Compendium, 1997).
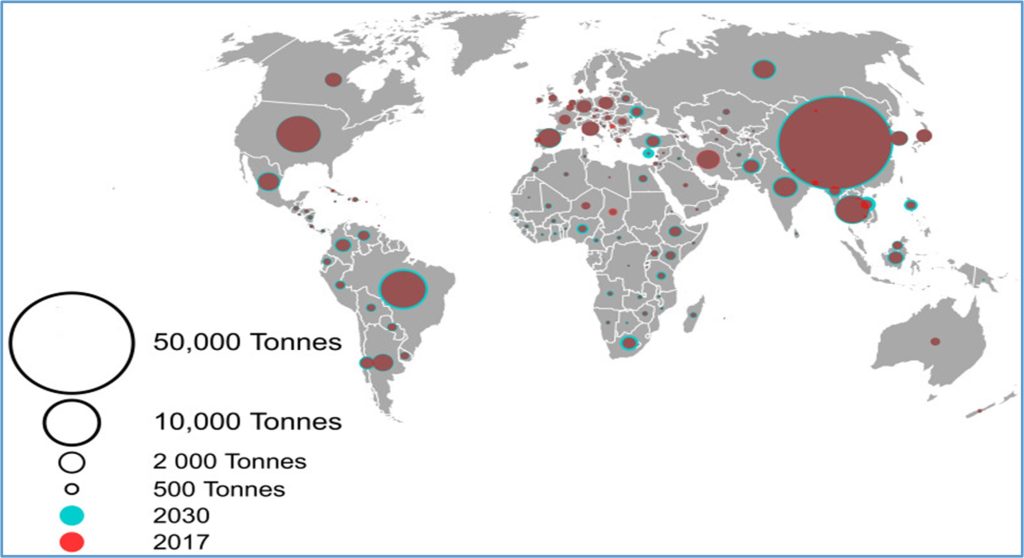
The use of antibiotics in animal farming — a major contributor to antimicrobial resistance is expected to grow by 8% between 2020 and 2030 despite ongoing efforts to curtail their use, according to an analysis(fig:1)
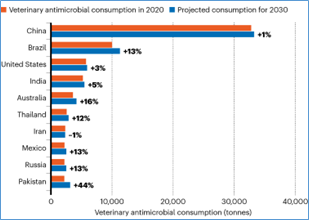
Table3. gives the classification and mode of action of commonly used antibiotics in animal treatment.
Table :3. Classification of Antibiotics and their mode of action
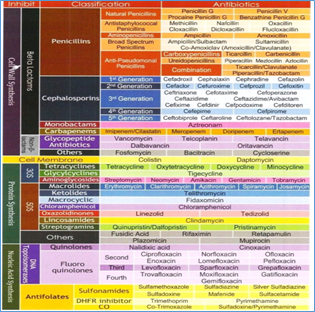
What is an Antibiotic Resistance(AMR)?
The World Health Organization defines AMR as “when bacteria, viruses, fungi, and parasites change over time and no longer respond to medicines, making infections harder to treat, and increasing the risk of disease spread, severe illness, and death. Antimicrobial resistance happens when the bacteria and fungi develop the ability to defeat the drugs designed to kill them. That means the bacteria are not killed and continue to grow(fig3). Resistant infections can be difficult, and sometimes impossible, to treat
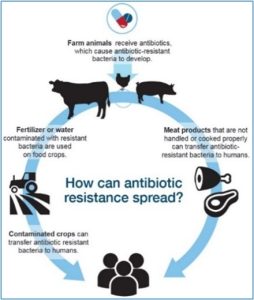
Fig:3. Development of Antibiotic Resistance (Source: CIFE Science Fact Sheet)
Antibiotic Misuse/abuse/ over use cause AMR
Over-prescription of drugs, use of too high or too low dosages, and incorrect duration of medication can aggravate the problem. Antibiotic misuse, sometimes called antibiotic abuse or antibiotic overuse, refers to the misuse or overuse of antibiotics, with potentially serious effects on health. It is a contributing factor to the development of antibiotic resistance, including the creation of multidrug-resistant bacteria, informally called “super bugs“: such as E.Coli, Staphylococcus, Enterococcus and Acinetobacter can develop resistance to multiple antibiotics and cause life-threatening infections.
Most of the times, the organisms get resistance to more than one antibiotic called as “Multidrug-resistant” (MDR) organisms. Infections caused by MDR organisms are associated with increased mortality compared to those caused by susceptible bacteria and they carry an important economic burden. These bacteria are able to cause serious disease and this is a major public health problem. Important examples are:
- Methicillin-resistant Staphylococcus aureus (MRSA)
- Vancomycin-resistant Enterococcus (VRE)
- Multi-drug-resistant Mycobacterium tuberculosis (MDR-TB)
- Carbapenem-resistant Enterobacteriaceae (CRE) gut bacteria.
How does antibiotic resistance occur?
Antibiotic resistance is a natural occurrence caused by mutations in bacterial genes. However, inappropriate use of antibiotics accelerates the emergence and spread of antibiotic-resistant bacteria. The primary driver for evolution of resistant genes is the excessive use of antimicrobials that exerts selection pressure and thus escalates the favourable mutation in the bacteria(fig4). Although antimicrobials are used in comparable amounts in animals and humans (118 mg/PCU and 133 mg/kg respectively), the chances of mutations in animals are higher due to larger animal biomass.
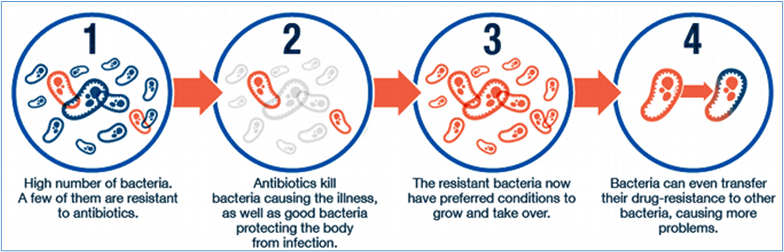
Fig:4. Occurrence of AMR (Source: The Pharma News)
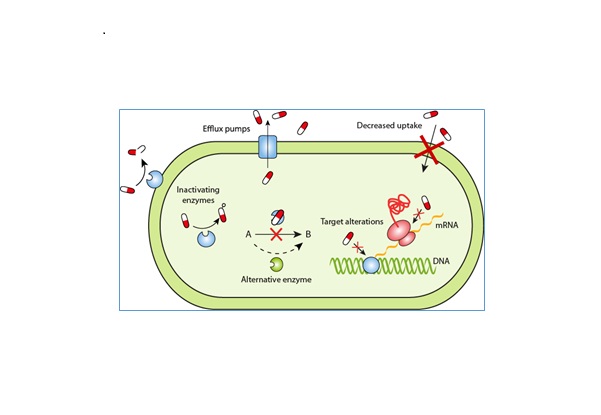
Mechanism of organisms become Resistant to Antibiotics
There are several general methods through which a cell can become resistance to an antibiotic. These mechanisms are:
- Decreased cell permeability to the drug – the cell can change its membrane structure so that the drug cannot enter the cell and perform its function
- Alter the drug binding/recognition site – by changing the structure of the membrane surface, the site which previously allowed the drug to bind to the cell can no longer do so
- Chemical modification of the antibiotic – by cleaving a portion of the molecule or adding a substituent group, the properties of the active molecule in the antibiotic can be altered such that it is rendered harmless to the cell
- Active transport – the transport of drug molecules out of the cell. In many cases, this is done via a drug/proton antiport system. With this mechanism, H+ ions are pumped into the cell as drug molecules are pumped out.
- Enzyme or pathway alteration – the cell can change the pathway or enzyme used to carry out a cell process occurs. By doing this, the cell can bypass the enzyme that is affected and cause the drugs effects to have no bearing on the functioning of the cell(fig5).
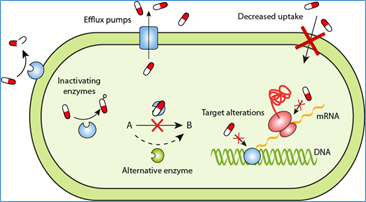
Fig:5. Antibiotic resistance strategies in bacteria. (Courtesy of E. Wistrand-Yuen).
Why is antibiotic resistance a major consumer health concern?
Antimicrobial resistance has drastically increased because of the global misuse and overuse of antibiotics. This is prompted by the high prevalence of foodborne zoonotic pathogens such as E. coli, Enterococcus spp., S. aureus, and non typhoidal Salmonella and Campylobacter spp.
Global monitoring epidemiology reported the resistance of E. coli and Salmonella strains to ampicillin, chloramphenicol, and trimethoprim-sulfamethoxazole. It is confirmed that the application of antimicrobials in food animals might lead to the development of resistant strains of bacteria, which propagates to infect both animals and humans.
Antimicrobial drug resistance of E. coli isolated from poultry abattoir workers at risk .It is also reported that the antimicrobial usage in food animals to be increasing and resulting in the development of multy drug drug–resistant bacteria. It has been suggested that this resistant bacterial strain can be transferred to people working with such animals, for example, abattoir workers.
Antimicrobial drug resistance was investigated for E. coli from broilers raised on feed supplemented with antimicrobials and the people who carry out evisceration, washing, and packing of intestines in a high-throughput poultry abattoir. In addition to their specific effects, edible tissues of poultry might contain veterinary drug residues, which would cause hazardous health effects in humans, such as toxicological effects, hypersensitivity, allergic reactions, change of gut microflora, and increased bacterial resistance to antimicrobials. Serious concerns are raised on the antimicrobial resistance in zoonotic enteropathogens (Salmonella spp., Campylobacter spp.), commensal bacteria (E. coli, enterococci), and bacterial pathogens of animals (Pasteurella, Actinobacillus spp.). Recent studies indicate that the antibiotics used in livestock can affect the soil microenvironment and even climate,
Therefore, AMR is regarded as one of the predominant and most salient public health issues of the 21st century, as it threatens the effective treatment and prevention of an ever-growing range of infections caused by bacteria, viruses, fungi, and parasites. These may be harder to treat than those caused by non-resistant bacteria. The overuse of antibiotics in recent years means they’re becoming less effective and has led to the emergence of “superbugs”. These are strains of bacteria that have developed resistance to many different types of antibiotics, including: MRSA (methicillin-resistant Staphylococcus aureus, Clostridium difficile). Antibiotic resistance leads to higher medical costs, prolonged hospital stays, and increased mortality.
Table :4. Presence of antibiotic residues in the different animal-derived products and their consequences in humans and animals. (Source: Compiled and edited by Dr Abhinav Gaurav, Program Director, Bihar Sustainable Livelihood Development (BSLD) Program)
| N | Antibiotic residues | Consequences in Humans/Animals |
| 1 | Tetracyclines | Primary and permanent teeth discolouration in children and infants, allergic reactions, and teratogenicity during the first trimester of pregnancy, nephrotoxicity, carcinogenic, hepatoxicity, and disturbance of the normal microflora of the intestines. It equally causes skin hyperpigmentation of areas exposed to the sun, proximal and distal renal tubular acidosis, hypersensitivity reactions. |
| 2 | Oxytetracyclines | Carcinogenicity and cytotoxicity in bone marrow of broiler chickens |
| 3 | Sulfadimidine | Carcinogenicity and allergic reactions. |
| Sulfamethoxazole | Carcinogenicity and allergic reactions | |
| 4 | Sulphamethazine | Carcinogenicity and allergic reactions |
| 5 | Enrofloxacin | Allergic hypersensitivity reactions or toxic effects, phototoxic skin reactions, chondrotoxic), and tendon rupture. |
| 6 | Chloramphenicol | Bone marrow toxicity, optic neuropathy, brain abscess |
| 7 | Penicillin | Allergy, affect starter cultures to produce fermented milk product |
| 8 | Quinolones | Allergic hypersensitivity reactions or toxic effects (phototoxic skin reactions, chondrotoxic) and tendon rupture. |
| 9 | Amoxicillin | Carcinogenic, teratogenic, and mutagenic effects. |
| 10 | Penicillin G | Allergy (hypersensitivity reaction) ranging from mild skin rash to life-threatening anaphylaxis. |
Ban of Antibiotic Use in Animals
Resistant bacteria in food can cause infections in humans. Similar to humans, giving antibiotics to food animals will kill most bacteria, but resistant bacteria can survive. When food animals are slaughtered and processed, resistant germs in the animal gut can contaminate the meat or other animal products. Resistant germs from the animal gut can also get into the environment, like water and soil, from animal manure. If animal manure or water containing resistant germs are used on fruits, vegetables, or other produce as fertilizer or irrigation, then this can spread resistant germs. Many countries already ban growth promotion antibiotics and the European Union has banned the use of antibiotics for growth promotion since 2006. On January 1, 2017, the FDA enacted legislation to require that all human medically important feed-grade antibiotics (many prior over-the-counter-drugs) become classified as Veterinary Feed Directive drugs (VFD). This action requires that farmers establish and work with veterinaries to receive a written VFD order. The effect of this act places a requirement on an established veterinarian-client-patient relationship (VCPR). Through this relationship, farmers will receive an increased education in the form of advice and guidance from their veterinarian.
Which antibiotic is banned in EU Countries and in India for use in animals?
The use of antibiotics for growth promotion purposes was banned in the European Union from 2006, and the use of sub-therapeutic doses of medically important antibiotics in animal feed and water to promote growth and improve feed efficiency became illegal in the United States on 1 January 2017, through regulatory change. Even as the EU has put an end to non-therapeutic antibiotic use, things have not moved much in India. There have only been deliberations and no action to restrict Antibiotic Growth Promoters (AGP) in feed, that is widely available with manufacturers, veterinary medicine dealers, or even sold online. Although the Bureau of Indian Standards (BIS) recommends not using antibiotics with systemic action as AGPs in feed, this is not mandatory. An expert panel set up by the BIS to make the restriction mandatory has not taken significant action so far.
In India Colistin resistance is increasing in clinical practice and Celestin is extensively used in poultry and aqua farming as a growth promoting agent”, and such practice should stop
Follow withdrawal period
The purpose of the withdrawal periods is to ensure that foods do not contain residues of pharmacologically active substances in excess of the maximum residue limit (MRL). To determine withdrawal periods, data on the drug metabolism in the animal is studied.
Withdrawal period refers to the minimum period of time from administering the last dose of medication and the production of meat or other animal-derived products for food. Animals treated with a product that has a withdrawal period of 45 days should be withheld from sale or slaughter for at least 45 days. Withdrawal periods are not the same for all drugs. Some withdrawal periods are very short, 1 or 2 days. Some are longer, 7-10 days. Some are even longer, up to weeks.
Table :5. Withdrawal time and tolerance level of antibiotics in chickens
| Drugs | Pre-slaughter
withdrawal time (days) |
Tolerance level (ppm) |
| Bacitracin | 0 | 0.05 |
| Chlortetracycline | 1 | 0 |
| Erythromycin | 2 | 0.125 |
| Gentamycin | 35 | – |
| Streptomycin | 4 | 0.10 |
| Carbomycine | 1 | 0 |
| Monensine | 5 | 0.05 |
Table:6. Recommended withdrawal periods for some of the coccidiostats and antimicrobial substances used in poultry industry
| Antimicrobials | Withdrawal period ( in days) |
| Levamisole | 7 |
| Nicarbazin Narasin | 5 |
| Ciprofloxacin | 15-19 |
| Monensin | 3 |
| Oxytetracycline Sulphadimine | 5 |
| Ampicillin | 6 |
Conclusion
Drug resistance is one of the major concern globally. This will cause a grave impact on treatment of illness caused by bacteria. The use of antibiotics for growth promotion purposes was banned in many countries, and the use of sub-therapeutic doses of medically important antibiotics in animal feed and water to promote growth and improve feed efficiency. In future, much of the effort to control pathogens in food animals, including poultry, will depend on use and prompt withdraw and the increased use of vaccination programs. Advances in molecular biology will permit specific pathogen antigens to be cloned and synthesized, facilitating their use in vaccines to stimulate the animal’s own natural defense mechanisms. Production of antibiotic free animals and poultry and development of alternatives to antibiotics will be the future trend and research.
(Acknowledgement: The author duly acknowledges Google reference s and illustrations)


