Colibacillosis In Poultry: An Overview

Dr. Kuldip Joshi (M.V.Sc. Scholar), Dr. C. J. Dave (Assistant Professor), Dr. D. J. Ghodasara (Professor),
Dr. Digjay Kabariya (PhD. Scholar), Dr. Devasi Solanki (M.V.Sc. Scholar), Department of Veterinary Pathology,
College of Veterinary Science and Animal husbandry, Anand-388001, Kamdhenu University, Gujarat
❖ Introduction:
Colibacillosis is a localized or systemic bacterial infection of avian species. It is most frequently observed in chicken and turkey. Colibacillosis is caused by Avian Pathogenic Escherichia coli (APEC) bacteria which belongs to the enterobacteriaceae family. It is manifested in various forms such as acute septicemic form which is called colisepticemia, yolk sac infections in chicks (omphalitis/mushy chick disease), coligranuloma (Hjarre’s disease), swollen head syndrome, avian cellulitis, salpingitis, egg peritonitis, osteomyelitis/synovitis, and pantophthalmitis.
E. coli bacteria are normal inhabitants of the gastrointestinal tract of birds and most of the strains are non-pathogenic in nature, so they are widely distributed in the faeces, so birds are continuously exposed to it through contaminated water, litter material, utensils and farm workers. Presence of rodents, insects and flies in the farm is also a risk factor which contributes to the spread of colibacillosis. Colibacillosis is responsible for the elevated morbidity and mortality which leads to heavy economic losses on a farm especially around the peak of egg production and throughout the late lay period (Linden, 2015). In addition, E. coli can enter the egg through the penetration of the egg shell when the egg is contaminated by feces, contaminated hatchery equipment. It can infect the chicks during hatching, which leads to high early chick mortality and infection of the yolk sac (Kabir, 2010). Although all age groups of birds are susceptible the younger birds show severe form of disease than older birds. Colibacillosis is most commonly seen with other diseases such as Mycoplasmosis/CRD which causes combined infection known as CCRD (complicated chronic respiratory disease), Infectious bursal disease (IBD), Infectious bronchitis (IB), Newcastle disease (ND), and sometimes with coccidiosis. Due to its occurrence with other diseases and mixed infection, it is difficult to diagnose colibacillosis early in the course of disease.
❖ Etiology :
Colibacillosis is caused by various strains of Avian Pathogenic Escherichia coli (APEC). Most commonly involved strains of E. coli are O1, O2, O18 and O78 (Wang et al., 2014). Escherichia coli is a gram-negative, non-acid fast, non-spore-forming rods of up to 3 μm in length. They come under the Enterobacteriaceae family of bacteria and are motile with peritrichous flagella.
E. coli has many virulence-associated factors, which includes adhesins, toxins, iron acquisition factors, lipopolysaccharides, polysaccharide capsules, and invasins (Sarowska et al., 2019)
❖ Source of infection:
E. coli is ubiquitous in nature and cosmopolitan in distribution. It is a common inhabitant of the intestinal tract of poultry and other birds. The feed, water, litter, faecal material, dust particles inside poultry shed, rodent droppings are the source of infection. Transmission is also possible from breeder/layer to the new born chick via vertical route (Petersen, 2006)
❖ Predisposing factors:
Rearing of birds of different age group together, exposure to bacterial disease such as Mycoplasma, and viral diseases such as IBD, IB, ND, poor ventilation, overcrowding, excessive dust and ammonia concentrations, stress of production in young layer birds, vent trauma due to excessive size of egg, vent prolapse or vent cannibalism. Chicks less than 10 days of age are particularly susceptible for the infection.
❖ Route of transmission:
E. coli infection in poultry can occur with various route which includes,
1) Respiratory route: The inhalation of contaminated dust of poultry houses is the most common means of infection in poultry. The damage to respiratory tract mucosa from diseases such as IB, Mycoplasmosis may predispose to the infection of E. coli. The excessive amount of dust and ammonia can predispose to infection of E. coli due to the irritation they cause on the respiratory tract mucosa. High NH3 levels (>25 ppm) damage bird’s respiratory systems and its mucous lining, thereby increasing their susceptibility to respiratory infections, particularly to E. coli infection (Swelum et al., 2021)
2) Gastrointestinal tract: As E. coli is normally present in the gastrointestinal tract, any damage to the mucosa of the GIT can lead to the entrance of the organism in the blood and subsequent septicaemia. Coccidiosis, general enteritis, haphazard use of antibiotics, mycotoxins, poor water and feed quality, abrupt changes in feed may disrupt the normal balance of gut microflora and cause pathogenic E. coli to flare up and invade the gut. Other than this contaminated water and feed may act as a source of infection via gastrointestinal route.
3) Reproductive tract: Ascending infection of E. coli is also possible via the vent of birds. The vent of birds can be damaged either due to prolapse, vent pecking or larger egg size. The ascending infection of the E. coli can happen due to the common opening of the digestive and reproductive tract in the cloaca. The major factors responsible for vent prolapse include intestinal infection, overweight in birds, age at sexual maturity, early photostimulation, unbalanced feed formulation and laying double yolked eggs with larger size (Ray et al., 2013). The involvement of hormonal factors is also documented. The bird affected with vent prolapse show decreased levels of 17 alpha- estradiol than unaffected birds (Shemesh et al., 1984)
4) Skin: The damaged skin of birds may cause the infection of E. coli via contaminated dust and litter material and faecal matter.
❖ Clinical forms:
Colibacillosis is seen in two forms in poultry:
1) Systemic form and 2) Localized form
– Systemic forms of Colibacillosis
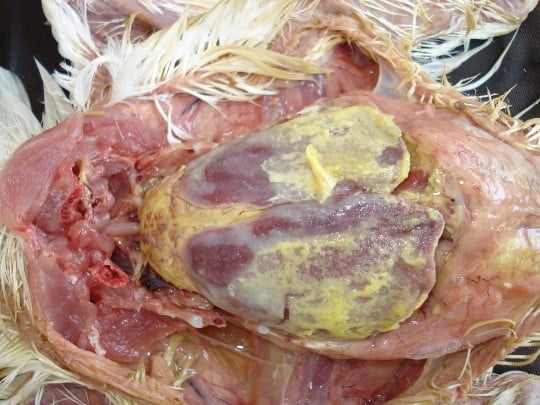
Colisepticemia: Colisepticemia is characterized by the presence of E. coli in the bloodstream of the birds. The severity of the disease depends on the virulence of strain, number of organisms contracted, age of birds, stress factors etc. Colisepticemia is characterized by initial respiratory infection (airsaculitis) and then followed by the generalized infection including perihepatitis, pericarditis and septicemia (Mellata et al., 2003).
The nature of the exudate covering the heart and peritoneum is fibrinous type. The respiratory tract involvement may cause signs such as respiratory rale, sneezing, cough and gasping for air. The septicemia may lead to osteomyelitis, tenosynovitis, and granuloma formation.
On postmortem examination, presence of yellowish cheesy exudate covering the heart and liver in the form of fibrinous pericarditis and perihepatitis is evident in sever cases. In mild cases airsaculitis with some cheesy yellowish plaque formation and clouding of airsacs is evident.
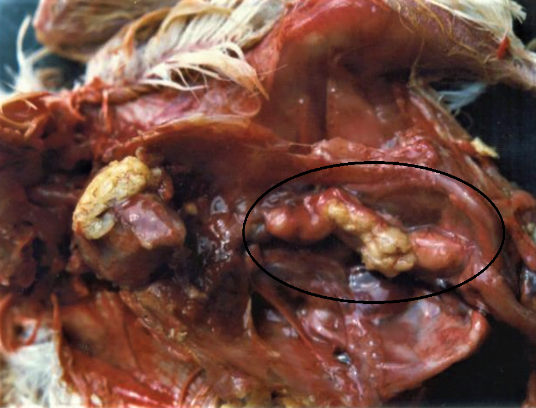
Coligranuloma: this form of colibacillosis is also called Hjarre’s disease and it is a sporadic form of infection. This form of disease is characterized by formation of multiple granuloma in intestine, uterus, mesentery and in liver (Islam et al., 2007).
Localized forms of colibacillosis
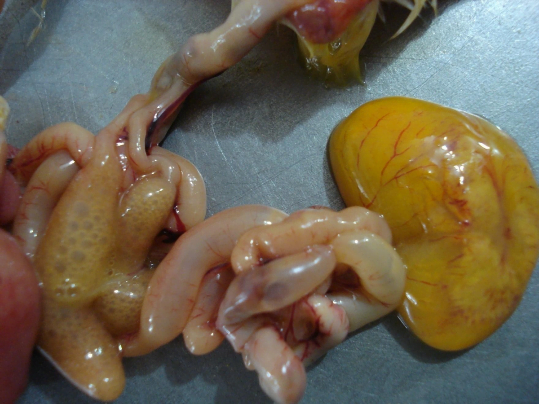
Yolk sac infection/ Omphalitis: it is the most common form of localized colibacillosis and it affects young chicks. It happens due to poor hygienic conditions in hatcheries and contamination of egg shell with E. coli which leads to omphalitis of the newborn and subsequently infection of adjacent yolk sac. It is the major cause of early chick mortality. Affected chicks show signs of depression, swollen abdomen and tendency to huddle. On postmortem, distended foul smelling unabsorbed yolk can be seen, yolk loses its normal shiny golden yellow appearance and becomes granular and curdled in appearance. The carcass is often dehydrated and congested. The tissue around the navel is often moist and congested, so it is called Mushy chick disease.
Yolk sac infection may be multietiology in nature. E. coli, Staphylococcus aureus, Proteus mirabilis are commonly isolated bacteria from the case of yolk sac infection in which E. coli is most commonly isolated bacteria (Amare et al., 2013)
Swollen head syndrome and coliform cellulitis: swollen head syndrome is an acute to subacute form of localized colibacillosis in which there is cellulitis of subcutaneous tissue affecting the periorbital areas of the face of chicken giving It name swollen head syndrome. swollen head syndrome is a multietiology disease where the initial lesions are caused by Avian metapneumovirus (aMPV), and the clinical signs are due to bacterial complications. The bacteria isolated from the swollen head syndrome affected birds include Escherichia coli, Staphylococcus aureus, Proteus spp., Corynebacterium spp., In which E. coli are the most frequently isolated bacteria (Momin et al., 2020). In coliform cellulitis the subcutis of the abdomen, thigh and midline are most commonly affected and there is formation of serosanguinous plaques on the surface of skin of affected portion.
Salpingitis and peritonitis: It is inflammation of oviduct of bird which results due to infection of oviduct by E. coli. Peritonitis (Inflammation of peritoneum) can also happen after the bacteria spread to peritoneum of the affected bird. Salpingitis results in reduced egg production, with death of laying hens and breeders sporadically. The oviduct is presented with a mass or multiple mass of caseous exudate with firm consistency when postmortem examination is performed. Other bacteria isolated from the case of salpingitis-peritonitis are staphylococci, Mannheimia haemolytica, and Streptococcus bovis (Jordan et al., 2005)
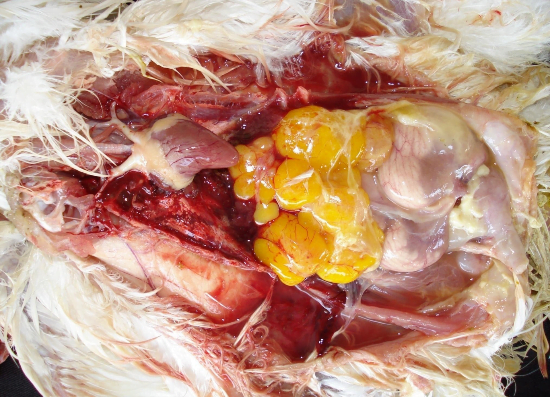
Egg peritonitis: it is inflammation of the peritoneum with the appearance of broken egg in the abdominal cavity which is characterized by fibrin or albumen-like material with a cooked appearance among the abdominal viscera and milky fluid in the abdominal cavity. It is the cause of sporadic death in layers or breeder hens, but in some flocks may become the major reason of mortality before or after reaching peak production (Rodrigo, 2019). Egg peritonitis is also seen in some other diseases, most commonly in LPAI.
❖ Diagnosis:
Colibacillosis is suspected based on the clinical features and the typical gross lesions at the postmortem. The diagnosis of E. coli can be done by its isolation from cardiac blood and affected tissues, like liver, and spleen. The cultural characteristic of E. coli on various agar are bright pink colonies on MacConkey agar, yellowish green colonies surrounded by an intense yellow green zone on BG agar and characteristic metallic sheen colonies on the EMB agar (Zinnah, 2007). Further confirmation can be done by PCR.
❖ Prevention and control:
Prevention of colibacillosis is best achieved by good sanitary and hygienic managemental practices. Source of infection and predisposing factors should be ruled out and should be eliminated to subside the ongoing disease. Faecal Contamination of eggs can be prevented by frequent egg collection, keeping the nest material clean, discarding the broken eggs properly and disinfecting the eggs within 2 hours of laying. Outbreak of colibacillosis has been controlled by chlorination of water and the use of closed irrigation systems (Dhillon & Jack, 1996). Maintaining proper ventilation and litter quality is a very important element in reducing the risk of development of colibacillosis. Proper ventilation will ensure the decrease in dust and ammonia accumulation, which will help to prevent respiratory mucosal damage. The control of rodents and other insects is also of importance for prevention of colibacillosis. Strict biosecurity measures should be adopted, which includes sanitation of housing, instruments and hygiene of workers. Regular vaccination of birds for immunosuppressive disease like IBD and procurement of Mycoplasma free eggs. Other than that, nutritional management such as provision of vitamin A, vitamin E, selenium and probiotics can improve the overall health of the birds and help in reducing the incidence of colibacillosis (Linden, 2015). Several inactivated or live attenuated vaccines have been tested experimentally, and are proven to be beneficial for preventing Colibacillosis and are available commercially for mass vaccination of poultry.
❖ Commonly observed forms of colibacillosis with their characteristic postmortem lesion:
References :
Amare, A., Amin, A. M., Shiferaw, A., Nazir, S., & Negussie, H. (2013). Yolk sac infection (omphalitis) in Kombolcha poultry farm, Ethiopia. American-Eurasian Journal of Scientific Research, 8(1), 10-14.
Dhillon, A. S., & Jack, O. K. (1996). Two outbreaks of colibacillosis in commercial caged layers. Avian Diseases, 40, 742–746. DOI: 10.2307/1592290
Islam, M. N., Fatema, B. Z., & Faruk, M. I. (2007). Hjarreâ s disease in chickens: clinical, pathological, microbiological and therapeutic findings. Bangladesh Journal of Veterinary Medicine, 49-53
Jordan, F. T. W., Williams, N. J., Wattret, A., & Jones, T. (2005). Observations on salpingitis, peritonitis and salpingoperitonitis in a layer breeder flock. Veterinary record, 157(19), 573-577.
Kabir, S. M. L. (2010). Avian Colibacillosis and Salmonellosis: A Closer Look at Epidemiology, Pathogenesis, Diagnosis, Control and Public Health Concerns. International Journal of Environmental Research and Public Health, 7, 89-114. doi:10.3390/ijerph7010089
Linden, J. (2015). Colibacillosis in Layers: an Overview. The Poultry Site. Retrieved from http://www.thepoultrysite.com/articles/3378/colibacillosis-in-layers-an-overview/ on 18th may,2021
Mellata, M., Dho-Moulin, M., Dozois, C. M., Curtis, R., Lehous, B., & Fairbrother, J. M. (2003). Role of avian pathogenic Escherichia coli virulence factors in bacterial interaction with chicken heterophils and macrophages. Infection and Immunity, 71(1), 494-503. Doi 10.1128/IAI.71.1.494503.2003f
Momin, S.G., Ghodasara, D.J., Mathakiya, R.A., Dave, C.J., Jani, P.B., Kabariya, D.V., & Pradhnya Choudhari1 (2020). Cultural Isolation and Identification of Possible Causative Agents of Swollen Head Syndrome in Broilers. Ind J Vet Sci and Biotech, 16(2,3, &4): 86-88.
Petersen, A., Christensen, J.P., Kuhnert, P., Bisgaard, M. & Olsen, J.E. (2006). Vertical transmission of a fluoroquinolone-resistant Escherichia coli within an integrated broiler operation. Veterinary Microbiology, 116, 120–128. 10.1016/j.vetmic.2006.03.015
RAY, S., SWAIN, P. S., AMIN, R. U., NAHAK, A. K., SAHOO, S. K., RAUTRAY, A. K., & MISHRA, A. (2013). PROLAPSE IN LAYING HENS: ITS PATHOPHYSIOLOGY AND MANAGEMENT: A REVIEW. Indian J. Anim. Prod. Mgmt. Vol, 29(3-4), 17-24.
Rodrigo A. Espinosa (2019). Egg Peritonitis in Poultry (Egg yolk peritonitis). Retrieved from https://www.merckvetmanual.com/poultry/disorders-of-the-reproductive-system/eggperitonitis-inpoultry on 18th may,2021.
Sarowska, J., Futoma-Koloch, B., Jama-Kmiecik, A., & Frej-Madrzak, M. (2019). Ksi a zczyk, M.; Bugla-Ploskonska, G.; Choroszy-Krol, I. Virulence factors, prevalence and potential transmission of extraintestinal pathogenic Escherichia coli isolated from different sources: Recent reports. Gut Pathog, 11(10).
Swelum, A. A., El-Saadony, M. T., Abd El-Hack, M. E., Ghanima, M. M. A., Shukry, M., Alhotan, R. A., … & El-Tarabily, K. A. (2021). Ammonia emissions in poultry houses and microbial nitrification as a promising reduction strategy. Science of The Total Environment, 146978.
Shemesh, Mordechai & Shore, Laurence & Lavi, Sara & Ailenberg, M & Bendheim, U & Totach, A & Weisman, Y. (1984). The role of 17 beta-estradiol in the recovery from oviductal prolapse in layers. Poultry science. 63. 1638-43. 10.3382/ps.0631638.
Wang, S., Meng, Q., Dai, J., Han, X., Han, Y., Ding, C., … & Yu, S. (2014). Development of an allele-specific PCR assay for simultaneous sero-typing of avian pathogenic Escherichia coli predominant O1, O2, O18 and O78 strains. PLoS One, 9(5), e96904.
Zinnah, M. A., Bari, M. R., Islam, M. T., Hossain, M. T., Rahman, M. T., Haque, M. H., … & Islam, M. A. (2007). Characterization of Escherichia coli isolated from samples of different biological and environmental sources. Bangladesh Journal of Veterinary Medicine, 25-32




