THE IMPORTANCE OF MAINTAINING A HEALTHY GUT

Joaquim Boix ( Product Manager – Intestinal Health, ADIVETER )
 The gut is a complex system in which several factors act in order to achieve its optimal function. These factors that we could consider key are the immune system based on the local action of certain immunoglobulins – the intestinal morphology based on the enterocytes integrity and the unions between them -, the diet, and the whole of the microorganisms present – the microbiota .
The gut is a complex system in which several factors act in order to achieve its optimal function. These factors that we could consider key are the immune system based on the local action of certain immunoglobulins – the intestinal morphology based on the enterocytes integrity and the unions between them -, the diet, and the whole of the microorganisms present – the microbiota .
A good intestinal health relies on the balance between the saprophytic and pathogenic floras found in the gut. The breakdown of this balance is what is known as dysbiosis and can lead to disease when the saprophyte flora decreases or disappears and the pathogenic one increases. There are authors who consider dysbiosis as a breakdown of the balance between the microbiota and the host, considering the whole factors involved in the functioning of the digestive system. This perspective is given by the introduction of the concept of holobiont, in which the association of a macroorganism (animal or plant) and the microorganisms that make up its microbiota is considered as a single entity (Van de Guchte et al., 2018).
There is not a clear definition of what can be considered a healthy microbiota, even so, a high microbial diversity in the animal can be associated with a healthy situation (Ocejo et al., 2018).
The advances made in molecular biology allow, as of today, to carry out studies on the set of the genomes present in a given medium, the metagenome, without having to make the isolation and the culture of the different microorganisms, field that is dealt with by metagenomics. Thus, the set of genes of the microorganisms present in an animal (the microbiota) is called microbioma.
Several factors affect the microbiota composition, such as an excess of certain nutrients, a poor digestibility of them, the use of antibiotics and several environmental factors, as well as factors related to the animal itself (age, sex …).
The optimization of the microbiota characteristics has an obvious impact on the productive performance of the animals and consequently on their profitability.
Although there are few studies on this subject, there is evidence that the gut microbiota composition may partly explain differences in Feed Efficiency in pigs. Although microbial diversity doesn’t vary between animals, Feed Efficiency can be related to some specific intestinal microorganisms, so they could potentially be used as biomarkers of this Feed Efficiency and even consider the use of these microorganisms as a probiotic. For example, at de genus level it has been stated that there is a positive correlation between Prevotella and pig weight (McCormack et al., 2017)
MICROBIOTA
The distribution of microorganisms throughout the animal intestinal tract is not uniform. This microbial configuration varies according to the gut section considered, both qualitatively and quantitatively. For example, in the case of birds, a greater number and diversity of microorganisms is observed in the cecum in relation to sections of the small intestine (Ocejo et al., 2018), and in the case of pigs, diversity and composition vary between the small intestine and the large intestine (Crespo et al., 2017). The total number of bacteria in the pig colon is estimated to be between 1010-1011 ufc/g of gut content (Gaskins et al., 2002) and in the cecum of birds it is around 1010ufc/g of gut content, compared to the 103 ufc/g of the small intestine.
This high microbial diversity in the bird caecum is due to a greater intake retention and to the fermentations that occur in this gut section (Ocejo et al., 2018). In the case of pigs, reasons are similar: in the small intestine there is a shorter transit time of the digesta and a continuous entry of new bacteria coming from the feed (Donaldson et al., 2016).
Diversity measures of the microbial population.
Alpha Diversity: this diversity measure describes the number of different species in a given population and its structure, based on the calculation of a series of indices. The main indices used are:
– Observed Species: expresses the number of species present in a given environment.
– Chao 1: basically reflects the number of species present in a specific population based on the determination of presence-absence data, so they are an expression of richness.
– Shannon and Simpson: These estimators describe the structure of a population with respect to the proportional abundance of each species. The Shannon index considers the number of species present (richness) and the relative quantity of individuals of each one of these species (abundance). Regarding the Simpson Index, it also takes a certain number of species present in an environment and their relative abundance.
Beta Diversity: this measurement of diversity describes the variation in the structure of a given community, in a given environment, over time, or the difference between populations of different environments in a given time. The estimator used is the Bray-Curtis index that quantifies the dissimilarity between two environments based on the counts of each medium. It expresses whether two populations have the same composition or they don’t share any species.
Phylogenetic Diversity Measurements: The indices described above describe diversity considering all species in the same way without taking into account the evolutionary differences between them. There are other indices that also express diversity between populations based on the genealogic relationships between them, taking into account the different evolution models and expressing their uniqueness.
Taxonomy
The microbiota taxonomy of animals, as well as the average of certain microorganisms regarding the rest, might be a reflection of their health status.
There are three predominant phyla in the animal gut.
– Firmicutes: formed by Gram+ bacteria. It includes the families Lachnospiraceae, formed by butyric acid producing bacteria, Clostridiaceae and Lactobaciliaceae.
– Bacteroidetes: formed by Gram– bacteria. It includes the families Prevotellaceae and Rikenellaceae (Alistepes)
– Proteobacteria: it includes a wide variety of pathogenic microorganisms, such as Enterobacteriaceae; E. coli, Samonella, Vibrio, Neisseria.
Other bacterial families of interest and belonging to other phyla are the Bifidobateriaceae, belonging to the phylum Actinobacteria, and the Akkermansia genus (phylum Verrucomicrobia).
The intestinal flora varies with the age of the animal. The microbiota evolution throughout the whole live of the animal is influenced initially by the population provided by the parents and the environment and later by the feed source used. In the case of mammals the most evident change occurs during the shift from a diet based on milk to another one in which the main nutrients are other types of carbohydrates.
Variations in the microbiota have also been observed depending on the breed of the animals, so the genetic profile can be considered a predisposing factor of intestinal infection due to these changes in the microbiota (Guevarra et al., 2019). Other factors can affect the microbiota evolution such as environmental factors.
INTESTINAL HEALTH
As mentioned before there is not a definition of what can be considered a healthy microbiota, and consequently a correct intestinal health, although a series of mechanisms involved in the loose of the intestinal function can be considered, among them the appearance of dysbiosis, the loss of the intestinal barrier integrity and inflammation (Van de Guchte et al., 2018).
The challenge is presented when trying to predict the appearance of this loss of intestinal health in a predictive way, before the start of the problem, and not in a diagnostic way, once it appears. Hence the need to define which could be the useful biomarkers for this purpose.
Two groups of markers could be considered: microorganisms, that indicate a change in the microbiota (Microbial shift) and the host own molecules that reflect cellular inflammation, intestinal integrity breakdown or the epithelial cells condition. In human medicine, for example, faecal levels of calprotectin are assessed as a biomarker of intestinal inflammation.
Considered as indicators of good intestinal health, at the hindgut level, are the presence of butyrate producing bacteria, such as those belonging to the Lachnospiraceae and Ruminococcaceae families. In animals with intestinal challenge, a decrease of these families and an increase of Proteobacteria is observed. At small intestine level there are other bacteria of the Lachnospiraceae family, such as Candidatus Arthromitus, that are considered indicators of an optimal intestinal health.
It has been observed that piglets with diarrhoea, on day 38, showed a lower Simpson Index at day 7 than healthy piglets (Dou et al. 2017). The same study showed, when comparing the microbiota of these piglets with diarrhoea versus healthy piglets, that healthy piglets had a higher abundance of Prevotellaceae, Lachnospiraceae, Ruminococcaceae and Lactobacillaceae.
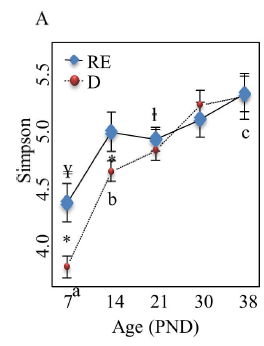
These remarks suggests that the intestinal microbiota affects the development of post weaning diarrhoea, and not only specific pathogens, and that it’s possible to differentiate diarrhoea susceptible piglets based on bacterial composition in early stages of piglet life (Dou et al. 2017)..
Regarding broiler, a correlation was observed between different taxa, specifically an appreciable decrease of the Lactobacillaceae family, associated to an increase of Campylobacter levels (Ocejo et al., 2018).
MICROBIOTA EVOLUTION
As mentioned before, the intestinal flora doesn’t remain constant throughout the animal life. This evolution that occurs over time can be influenced by many other factors, both environmental and genetic.
POULTRY
Broiler
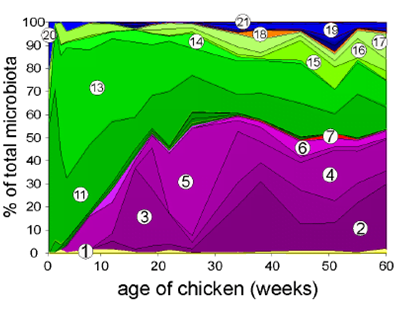
The intestine of chickens that come from the hatchery is not colonized by microorganisms from the hen, but by those from the environment. Initially, chicken microbiota is made up of facultative anaerobic bacteria. These bacteria consume oxygen and decrease its levels in the intestine, thus increasing the strict anaerobic bacteria population.
In one-day-old chicks, coming directly from the hatchery, the dominance of the phylum Proteobacteria is observed at caecal level, with a significant proportion of enterobacteria and E. coli among them. As the chick grows a decrease of this phylum is observed and the presence of Firmicutes and Bacteroides increases (Adiveter 2020).
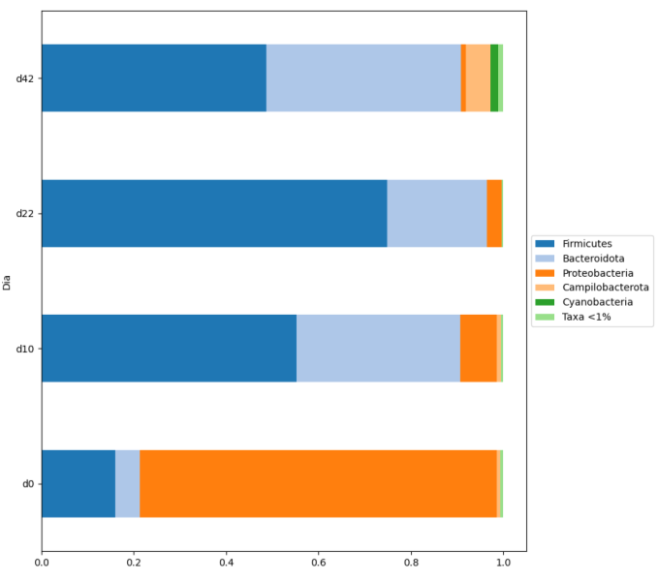
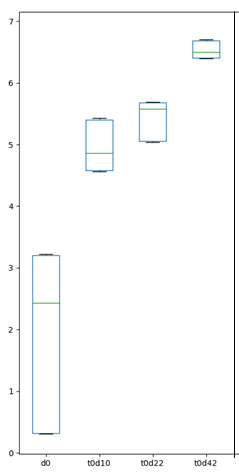
In a study carried out by Adiveter in a commercial farm, the values of the Shannon Index were determined at days 0, 10, 22 and 42; and an increase of this estimator was observed as the animals grew.
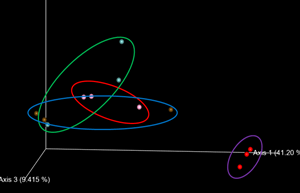
Regarding Beta Diversity, two different bacterial communities were observed according to age groups, showing that the bacteria of the one-day-old chicks are different from those of the rest of the animal life.
As it was observed, chicken rearing could be divided in three different stages. First days of chicken life in which Proteobacteria is the predominant phylum; a second stage, around 22 days of life, in which a clear decrease of proteobacteria and a significant increase in firmicutes is observed; and a third one, around 42 days of life, in which Bacteroidetes replaces part of the firmicutes.
Within Firmicutes, the predominant families are Lachnospiraceae and Ruminococcaceae that are butyrate producing bacteria. In the case of Bacteroidetes, the main genus are Bacteroides and Alistipes, mainly propionate producers (Qi et al, 2019; Polansky et al., 2015). Several authors explain this variation of the bacterial populations regarding the type of Volatile Fatty Acids produced. The production of butyrate could probably be associated to the high nutritional requirements of this early stage of the animal development and because it is a period of faster growth. In the case of propionate production it could be related to a balance between energy acquisition from available nutrients and a more sustained growth of the chickens (Ocejo et al., 2018).
(Ocejo et al., 2018) compared caecal microbiota of broilers and free range slow growing chickens, observing that free range chickens presented a higher proportion of gut health related bacteria, such as the Actinobacteria phylum of which bifidobacteria is part.
Even so, it must be taken into account that the microbiota profile of these chickens corresponds to animals that have never had contact with hens because they come from hatcheries. When adult hens are reared in contact with chickens, it has been observed that they act as donors of Bacteroidetes and Actinobacteria but do not act as an important source of Firmicutes. Chickens that were in contact with adult hens showed, in the firsts days of life, that about 40% of its caecal microbiota was made up of Bacteroidetes, compared to animals without contact with hens whose microbiota was dominated by Firmicutes (Kubasova et al., 2019).
Laying Hens
In studies in which the caecal microbiota of laying hens was determined, it was observed that the microbiota, until 24 weeks of age, was practically similar to that observed in chickens, reaching this age with the same trend of substitution of firmicutes, which were predominant, by Bacteroidetes.
The study stretched on until 7 months of age, observing that Firmicutes and Bacteroidetes l were still predominant and each one of them represented half of the total. The increase of Bacteroidetes, at the expenses of Firmicutes, was correlated with the increase of hen weight and egg production (Videnska et al., 2014).
PIGS
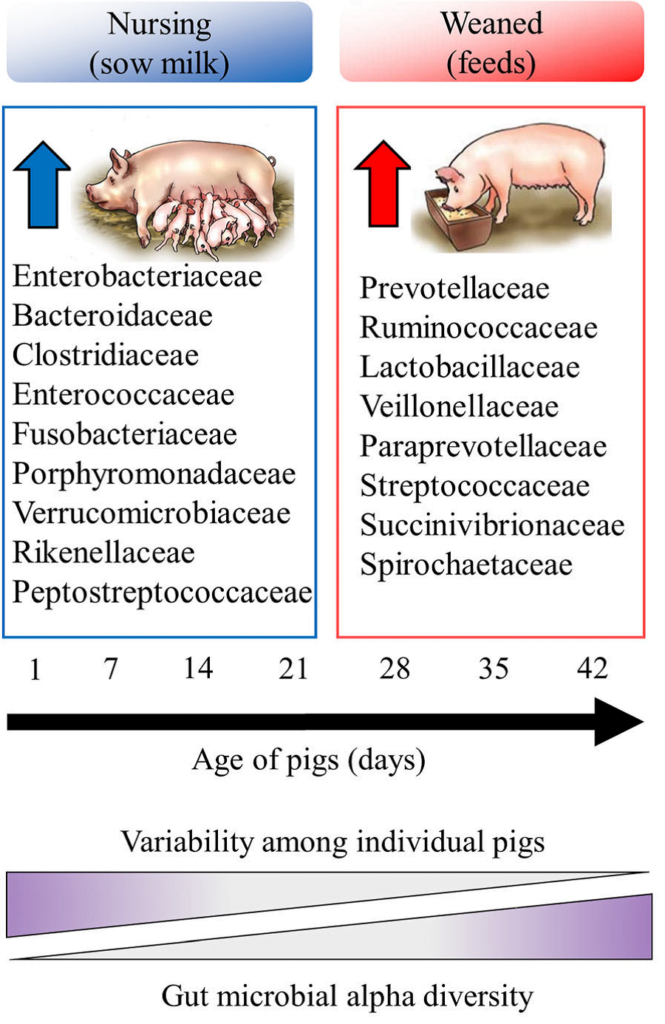
The pig microbiota colonization and evolution over time is hardly influenced by diet. The greatest change occurs in the weaning period with the shift from a breast milk based diet to another one based on solid feed (Nowland et al., 2019).
Studies have shown an increase in Alpha Diversity after weaning, and regarding Beta Diversity, significant differences between suckling and weaned piglets were also observed (Guevarra et al. 2019).
After weaning, an increase of Bacteroidetes phylum was observed, shifting from 44% in suckling piglets to 63% in the weaned ones. At the family level it goes from a population in which Ruminococcaceae (20%) and Prevotellaceae (13%) prevails in the case of suckling piglets, to another one with a higher proportion of Prevotellaceae and Lactobacillaceae, with increases of 44% and 4.8% respectively.
In the same way, Prevotella represents the main genus in both groups, increasing significantly from suckling piglets to the weaned ones. In the case of the weaned piglets, Prevotella and Lactobacillus were the predominant genus and in the case of the suckling piglets, Prevotella and Bacteroides (Guevarra et al. 2018).
These variations in the microbiota are mainly due to the type of diet the piglet receives. Bacteroides are microorganisms that use milk oligosaccharides as a source of energy and Prevotella is related to the fermentation of fibrous polysaccharides present in plants, as well as Lactobacillus that also metabolizes carbohydrates from plants, including starch (Guevarra et al. 2019).
MICROBIOTA MANIPULATION
As seen, gut microbiota is influenced by multitude of factors. The control of these factors establishes the profile of microorganisms that make it up.
Among the different elements that take part in the microbiota modulation, the following should be highlighted:
Environment
Microorganisms that colonize the intestinal tract have influence on the development of the immune system and hence on the health of the animal. The environment in which the animal develops itself affects this process and factors such as the hygienic state affects this development. Piglets reared together with their mother have greater flora diversity compared to their siblings weaned at 24 hours of age and reared in isolators (Inman et al., 2010).
Antibiotic use
When a group of piglets was subjected to a combination of antibiotics, a shift in the microbiota composition was observed after 14 days of treatment, compared to the animals not subjected to the medication. In animals subjected to the treatment, an increase in Proteobacteria was observed (from 1% to 11%), mostly E. coli which represented 62% of these microorganisms, besides a decrease of Bacteroidetes (Looft et al., 2012).
Feed processing.
The distribution of the microbial species in the gastrointestinal tract is also determined by the chemical composition and the structure of the digesta, since bacterial species differ in their substrate preferences and growth requirements.
It is known that particle size affects the digestibility of the diet and consequently their productive parameters. Likewise studies made at gastrointestinal microbiology level also demonstrated such an influence.
In the case of pigs, it is known that coarse particle size decrease gastric pH, thus maximizing the effectiveness of the “gastric barrier” against the transmission of faecal-oral pathogenic bacteria. Likewise, the low pH of the digesta ensures a higher proportion of Volatile Fatty Acids in its undissociated form, thus increasing its antimicrobial capacity. In addition to the gastric level effect, particle size also affects at cecum and colon levels, since it is possible that coarse particle size promote an increase of the population of Short Chain Fatty Acids-producing bacteria and consequently contributing to gastrointestinal health and preventing the proliferation of pathogenic bacteria (Kiarie et al., 2019).
In the case of poultry, it has been observed that the gizzard pH decreases when providing a coarse diet compared to a fine one due to an increase of the HCl secretion. Branton et al observed that birds fed with a coarse diet showed a lower mortality due to Necrotic Enteritis. This can be associated to the gastric function stimulation, including both HCl secretion and better utilization of nutrients in the small intestine that is related to coarse diets and influences the proliferation of C. perfringens (Engberg et al., 2002).
Hydrothermal treatments (pelleting, extrusion, expansion,…) affect the chemical structure of feed nutrients. In pelleted feed, part of the starch is retrograded and becomes resistant starch that can only be degraded by its fermentation by the large intestine microorganisms with the consequent production of short chain fatty acids. Likewise, pelleting alters the physicochemical characteristics of dietary fibre (De Vries et al., 2012). Even so, there are few studies on the effect of hydrothermal feed treatment on the microbial composition of the gastrointestinal tract (Kiarie et al. 2019).
Additives (prebiotics-probiotics)
Prebiotics and probiotics are additives often used in animal nutrition.
A prebiotic is a substance that without being hydrolysed or absorbed at gastrointestinal level selectively stimulates beneficial bacteria growth.
Probiotics are cultures of potentially beneficial bacteria for the intestinal flora and are administrated in order to colonize the large intestine and modify its microbiota.
The solution: Adifarm G+: An holistic approach to gram + bacteria control and a good intestinal health
Adifarm G+ is an exclusive and innovative formula based on resin acids and monolaurin that improves productivity and animal health thanks to the control of gram-positive bacteria in the gastrointestinal tract.
Resinic acids are free fatty acids of natural origin (conifer resins) with proven high efficacy against gram-positive bacteria.
• They produce a loss of the gram-positive bacteria cell membrane integrity and an increase in the permeability of protons and ions, causing cell lysis (San Feliciano et al. 1993, Sikkema et al 1995).
• They have effects on microbiota composition (Kettunen et al. 2015).
• They have anti-inflammatory effects and cell growth promoters.
Monolaurin is the esterified version of lauric acid with greater activity against gram + bacteria, presenting lower MIC than other acids and lauric acid.
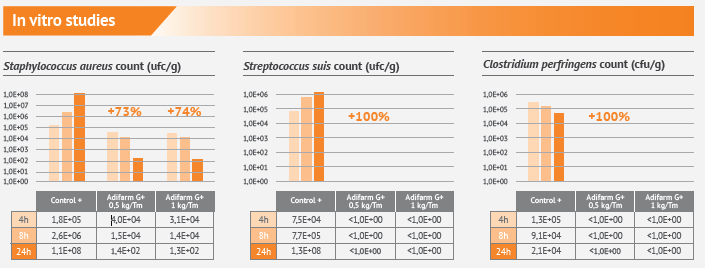
In vitro studies show a great efficacy against the main gram-positive bacteria affecting animal production.
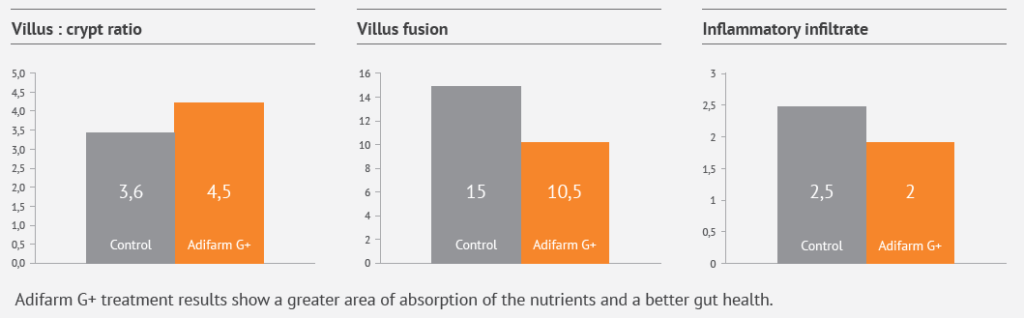
In vivo studies (histology): Adifarm G+ treatment results show a greater area of absorption of the nutrients and a better gut health:
In vitro studies (Metagenomics)
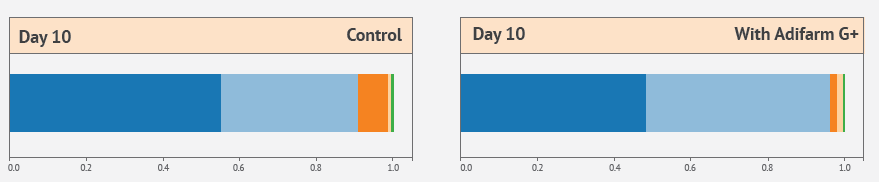
The normal evolution of the chicken cecum microbiota changes from a dominance of the phylum Proteobacteria (including Enterobacteriaceae), at the first stage of the animal’s life, to a progressive increase of Firmicutes. Later, Firmicutes decrease in favor of the phylum Bacteroidota. Treatment with Adifarm G+ shows a lower proportion of Proteobacteria at day 10 compared to the control treatment, so we can assume that the flora transition is faster.

The benefits of using Adifarm G+:
– High efficacy against gram-positive bacterias: Streptococcus suis, Clostridium perfringens.
– Controls pathogens and improves intestinal health to cope with reduced antibiotic production.
– Stable product against granulation heat stress (does not require microencapsulation to reach its effect in the entire gastrointestinal tract)
– Improves animal performance (increased digestibility and nutrient assimilation).
Adiveter Program for the control of intestinal microbiota
Gut microbiota is influenced by a wide variety of factors, and correlated with productivity. Our program allows a comprehensive approach to address specific pathogen control and maintain good intestinal health for the maximum animal performance.
1. Metagenomics study to know and understand gut microbiota health. Adiveter helps you to decipher the gut microbiota with advanced technology.
2. We design a tailor-made program according to results, species and productive stage to deal with gastrointestinal tract pathogens and maintain optimal productive performance.
BIBLIOGRAPHY
Dou, S.; Gadonna-Widehem, P.; Rome, V.; Hamoudi, D.; Rhazi, L.; Lakhal, L.; Larcher, T.; Bahi-Jaber, N.;Pinon-Quintana, A.; Guyonvarch, A. Characterisation of early-life fecal microbiota in susceptible and healthy pigs to post-weaning diarrhoea. PLoS ONE 2017, 12, e0169851.
Polansky O, Sekelova Z, Faldynova M, Sebkova A, Sisak F, Rychlik I. Important Metabolic Pathways and Biological Processes Expressed by Chicken Cecal Microbiota. Appl Environ Microbiol. 2015 Dec 28;82(5):1569-76. doi: 10.1128/AEM.03473-15. PMID: 26712550; PMCID: PMC4771310.
Qi Z, Shi S, Tu J, Li S. Comparative metagenomic sequencing analysis of cecum microbiotal diversity and function in broilers and layers. 3 Biotech. 2019; 9(8):316. doi:10.1007/s13205-019-1834-1.
Looft T, Johnson TA, Allen HK, Bayles DO, Alt DP, Stedtfeld RD, Sul WJ, Stedtfeld TM, Chai B, Cole JR, Hashsham SA, Tiedje JM, Stanton TB. In-feed antibiotic effects on the swine intestinal microbiome. Proc Natl Acad Sci U S A. 2012 Jan 31; 109(5):1691-6. doi: 10.1073/pnas.1120238109. Epub 2012 Jan 17. PMID: 22307632; PMCID: PMC3277147.
Kiarie EG, Mills A. Role of Feed Processing on Gut Health and Function in Pigs and Poultry: Conundrum of Optimal Particle Size and Hydrothermal Regimens. Front Vet Sci. 2019 Feb 19; 6:19. doi: 10.3389/fvets.2019.00019. PMID: 30838217; PMCID: PMC6390496.
Engberg RM, Hedemann MS, Jensen BB. The influence of grinding and pelleting of feed on the microbial composition and activity in the digestive tract of broiler chickens. Br Poult Sci. 2002 Sep; 43(4):569-79. doi: 10.1080/0007166022000004480. PMID: 12365514.
Van de Guchte, M., Blottière, H.M. & Doré, J. Humans as holobionts: implications for prevention and therapy. Microbiome. 2018 6, 81 https://doi.org/10.1186/s40168-018-0466-8
Gaskins HR, Collier CT, Anderson DB. Antibiotics as growth promotants: mode of action. Anim Biotechnol. 2002 May; 13(1):29-42. doi: 10.1081/ABIO-120005768. PMID: 12212942.
Donaldson GP, Lee SM, Mazmanian SK. Gut biogeography of the bacterial microbiota. Nat Rev Microbiol. 2016 Jan; 14(1):20-32. doi: 10.1038/nrmicro3552. Epub 2015 Oct 26. PMID: 26499895; PMCID: PMC4837114.
Ocejo, M., Oporto, B. & Hurtado, A. 16S rRNA amplicon sequencing characterization of caecal microbiome composition of broilers and free-range slow-growing chickens throughout their productive lifespan. Sci Rep 2019 9, 2506. https://doi.org/10.1038/s41598-019-39323-x.
Kubasova T, Kollarcikova M, Crhanova M, Karasova D, Cejkova D, Sebkova A, et al. Contact with adult hen affects development of caecal microbiota in newly hatched chicks. PLoS ONE 2019 14(3): e0212446. https://doi.org/10.1371/journal.pone.0212446
Ursula M. McCormack, Tânia Curião, Stefan G. Buzoianu, Maria L. Prieto, Tomas Ryan, Patrick Varley, Fiona Crispie, Elizabeth Magowan, Barbara U. Metzler-Zebeli, Donagh Berry, Orla O’Sullivan, Paul D. Cotter, Gillian E. Gardiner, Peadar G. Lawlor. Exploring a Possible Link between the Intestinal Microbiota and Feed Efficiency in Pigs. Applied and Environmental Microbiology 2017 Vol. 83, No. 15. https://orcid.org/0000-0002-5269-0387.
De Vries S, Pustjens AM, Schols HA, Hendriks WH, Gerrits WJ. Improving digestive utilization of fiber-rich feedstuffs in pigs and poultry by processing and enzyme technologies: a review. Anim Feed Sci Technol. 2012 178:123–38. doi: 10.1016/j.anifeedsci.2012.10.004.
Branton SL, Reece FN, Hagler WM Jr. Influence of a wheat diet on mortality of broiler chickens associated with necrotic enteritis. Poult Sci. 1987 Aug; 66(8):1326-30. doi: 10.3382/ps.0661326. PMID: 2891128.
Guevarra, R.B., Hong, S.H., Cho, J.H. et al. The dynamics of the piglet gut microbiome during the weaning transition in association with health and nutrition. J Animal Sci Biotechnol 2018, 9, 54. https://doi.org/10.1186/s40104-018-0269-6.
Guevarra RB, Lee JH, Lee SH, Seok MJ, Kim DW, Kang BN, Johnson TJ, Isaacson RE, Kim HB. Piglet gut microbial shifts early in life: causes and effects. J Anim Sci Biotechnol. 2019 Jan 14;10:1. doi: 10.1186/s40104-018-0308-3. PMID: 30651985; PMCID: PMC6330741.
Videnska P, Sedlar K, Lukac M, Faldynova M, Gerzova L, Cejkova D, et al. Succession and Replacement of Bacterial Populations in the Caecum of Egg Laying Hens over Their Whole Life. PLoS ONE 2014 9(12): e115142. https://doi.org/10.1371/journal.pone.0115142.
Nowland, T.L.; Plush, K.J.; Barton, M.; Kirkwood, R.N. Development and Function of the Intestinal Microbiome and Potential Implications for Pig Production. Animals 2019, 9, 76. https://doi.org/10.3390/ani9030076
Inman CF, Haverson K, Konstantinov SR, Jones PH, Harris C, Smidt H, Miller B, Bailey M, Stokes C. Rearing environment affects development of the immune system in neonates. Clin Exp Immunol. 2010 Jun;160 (3):431-9. doi: 10.1111/j.1365-2249.2010.04090.x. Epub 2010 Feb 22. PMID: 20184618; PMCID: PMC2883114.



