Colibacillossis: Threat to Poultry Health and Production
1*Nisar Ahmad Nisar, 2Shajhat Ahmad Qureshi, 3 Parveez Ahmad Para, 4Altaf Hussain Mir Pirzada,5 Ishfaq Ahmad Arawali Veterinary College, Sikar, Rajasthan-332 001.*Corresponding author: Nisar Ahmad Nisar, AVC, Sikar, Rajasthan 1, 4Arawali Veterinary College, Sikar, Rajasthan-332001 2Division of Veterinary Pathology , SKUAST-Jammu-181002 3Division of Livestock Products Technology, SKUAST-Jammu-181102 5Division of Animal Nutrition, SKUAST-Jammu-181102
The Indian poultry industry has come a long way – from a backyard activity to an organized, scientific and vibrant industry. The growth achieved so far is only a fraction of its immense potential. Poultry, which was considered as a backyard venture in the early 60’s has now been transformed into a strong agro based farming activity. Poultry occupies an important place in the Indian economy, as it contributes nearly Rs. 33000 crore (~2.5%) to the National GDP, gives employment to 30 lac persons directly or indirectly, earns 200 crores foreign exchange and produces 1.4 million tones of valuable organic manures. At present, annual growth rate is about 10 % in meat production and 6 % growth rate in egg production, total poultry meat production in 2009 was 3.6 million tons, while total egg production in 2009 was 61000 million.
Today, India ranks 5th in egg production and 11th in broiler production in the world. It is estimated that the poultry meat production is about 0.8 million tons per annum. Poultry utilizes substantial quantities of non-edible agricultural and industrial by-products and converts them into high quality nutritious protein rich food. It helps to bridge the gap between the requirement and availability of high quality protein for the human population of the country. This highly economical and productive venture is constantly threatened by various losses including infectious diseases that pose as a serious threat to the poultry industry (Yogaratnum, 1995), particularly in developing countries such as India.
Marked increase in poultry population along with the drastic changes in the husbandry practices resulted in an increase in the prevalence and variation in the pattern of the poultry diseases. Bacterial infections are important welfare issue for the chickens, but they are also economically important, because diseases are significant financial cost to the broiler industry, mainly due to mortality, growth retardation, uniformity reduction and condemnations ( Vandemaele et al., 2002; McKissick, 2006). Of the various diseases, colibacillosis is of prime importance. In the past few years, both the incidence and severity of colibacillosis have increased rapidly and current trends indicate that it is likely to continue and become an even greater problem in the poultry industry (Altekruse et al., 2002). Colibacillosis, which is caused by E. coli, causes considerable economic and welfare problems in broilers (Bettelheim, 1994; Vandemaele et al., 2002), due to its frequent occurrence and adverse effects on growth and health. Escherichia coli are normal inhabitant of the intestinal tracts of animals and birds and are harmless as long as it is kept in check by other intestinal bacteria (Barnes et al., 2003). When an imbalance occurs in bacterial flora of the intestinal tract, Escherichia coli may grow and cause an outbreak of colibacillosis. Clinical disease consists of respiratory signs, growth retardation, reduced feed intake and increased mortality (Vandemaele et al., 2002). Airsacculitis and fibrinous polyserositis (e.g. pericarditis, perihepatitis and peritonitis) are the main gross lesions, and septicaemia, sometimes acute, is also common (Whiteman et al., 1989). Enteritis caused by Escherichia coli (colibacillosis) is an important disease because of increased mortality and decreased performance. It is often considered as opportunistic pathogen because it infects whenever it has the opportunity to do so. Chickens of all ages are susceptible to colibacillosis, but usually young birds are considered more susceptible. Yolk infections in chicks and poults due to E. coli are primary infections from exposure to the bacterium during the hatching process. Egg shell contamination is the main reason for the exposure although poor hatcher sanitation can leave a residue of E. coli from the previous hatch. Losses normally occur during the first three days of life and present clinically as normal sized chicks on feed but dead. Lesions of caseous yolk material are often seen. Culture of this yolk material is used for diagnosis to differentiate from other possible bacterial causes such as Salmonella spp., Pseudomonas spp., Staph. aureus, etc. Some strains are associated with intestinal, respiratory, urinary or invasive infections. It can cause an infection under the skin known as cellulitis. Gross lesions in experimentally dead embryo included cranial and skin hemorrhages in addition to encephalomalacia in embryos Avian pathogenic E. coli (APEC) are often associated with other pathogens such as mycoplasma and viruses or with environmental hazards like dust and high concentration of ammonium in the air (Oyetunde et al., 1978 and Gross, 1994). The main route of entry is the ingestion into alimentary tract and via respiratory tract after the inhalation of faeces-contaminated dust. The intestine is the most important reservoir of APEC.
Clinical diseases are reported in chickens, turkey and ducks (Barnes et al., 2003). Natural infection occurs in quail (Zhou et al., 1995), pheasant (Swarbrick, 1985), pigeons (Schmidt et al., 2000), guinea fowl (Harpeet et al., 1993), ostriches (Welish et al., 1997) and emus (Hines et al., 1995). E. coli infection manifests itself in various clinical disease forms. The most frequent lesions in poultry (chickens, turkeys) associated with E. coli are systemic infections with lesions of airsaculitis, pericarditis, perihepatitis, peritonitis, septicaemia, synovitis, omphalitis and occasionally salphingitis and coligranuloma. The main route of entry is the ingestion into alimentary tract and via respiratory tract after the inhalation of faeces-contaminated dust. E. coli infections secondary to respiratory infections in broilers, egg-type pullets and layers, and turkeys, is an important disease scenario. The normal respiratory tract has the capability to remove E. coli and other bacteria from deep in the respiratory tract due to the action of ciliated epithelial cells lining the trachea. In respiratory disease situations such as Bordatella avium in turkey poults, infectious bronchitis in chickens, Mycoplasma gallisepticum infections in both turkeys and chickens, the epithelial cells of the trachea and bronchi of the lungs undergo changes that do not allow normal function of these cells. Hence E. coli numbers increase and infection of the respiratory tract occurs. The initial lesion seen when E. coli infection is established is a caseous airsacculitis. The deadly and chronic lesion seen with E. coli infection is a caseous peritonitis, perihepatitis, and/or pericarditis. House management practices can play a role in the incidence and severity of E. coli infections. Low ventilation rates that lead to high atmospheric ammonia, increased litter moisture, and high dust counts in the air all will aid in carrying more E. coli deep into the respiratory tissues. Increased litter moisture also has the effect of increasing the survivability and reproduction of E. coli bacteria. Egg layer colibacillosis is viewed as a primary disease due to E. coli. It is most often seen at the onset of egg production but can be seen at older ages as well. The sources of infection appear to be water, dust, or fecal material pecked off equipment. The stress of recent movement and the onset of egg production appear to be factors in the onset of the disease early in lay. One theory is that the young layer’s everted reproductive tract during egg laying is quite susceptible to infection with E. coli and the bacterium infects the bird by moving retrograde up the oviduct. Turkey poults suffer an E. coli related syndrome after viral enteritis. This is due to overgrowth of E. coli in the intestine from poor digestion of nutrients and a buildup of these undigested nutrients in the lower intestine. Poult death losses can be quite high with lesions seen as a septicemia or caseous peritonitis, perihepatitis, and/or pericarditis. Enteritis and diarrhoea caused by E. coli is associated with six pathotypes, each with its own different pathogenic mechanisms. They are Enteropathogenic E. coli, Enterohemorrhagic E. coli, Enterotoxigenic E. coli, Enteroaggrevative E. coli, Enteroinvasive E. coli and diffusely adherent E. coli (Donnenberg, 2005).
1. Enterotoxigenic E. coli (ETEC): ETEC strains are non-invasive, and they do not leave the intestinal lumen. It uses fimbrial adhesins (projections from the bacterial cell surface) to bind enterocyte cells in the small intestine. ETEC can produce two proteinaceous enterotoxins: the larger of the two proteins (LT enterotoxins) is similar to cholera toxin in structure and function, while the smaller protein (ST enterotoxins) causes cGMP accumulation in the target cells and a subsequent secretion of fluid and electrolytes into the intestinal lumen.
2. Enteropathogenic E. coli (EPEC): Like ETEC, they also cause diarrhoea, but the molecular mechanisms of colonization and aetiology are different. EPEC lack fimbriae, ST and LT toxins, but they utilize adhesin known as intimin to bind host intestinal cells. This verotype has an array of virulence factors that are similar to those found in Shigella, and may possess a shiga-toxin. Adherence to the intestinal mucosa causes a rearrangement of actin in the host cell, causing significant deformation. EPEC cells are moderately-invasive (i.e. they enter host cells) and elicit an inflammatory response. A change in intestinal cell ultra-structure due to “attachment and effacement” is likely the prime cause of diarrhoea in those afflicted with EPEC.
3. Enteroinvasive E. coli (EIEC): Infection causes a syndrome that is identical to Shigellosis, with profuse diarrhoea and high fever.
4. Enterohemorrhagic E. coli (EHEC): The most famous member of this verotype is strain O157:H7, which causes bloody diarrhoea and no fever. EHEC can cause Haemolytic-Uremic syndrome and sudden kidney failure. It uses bacterial fimbriae for attachment (E. coli common pilus, ECP), is moderately-invasive, and possesses a phage-encoded Shiga toxin that can elicit an intense inflammatory response.
5. Enteroaggregative E. coli (EAEC): So named because they have fimbriae which aggregate tissue culture cells, EAEC bind to the intestinal mucosa to cause watery diarrhoea without fever. EAEC are non-invasive. They produce a hemolysin and an ST enterotoxin similar to that of ETEC.
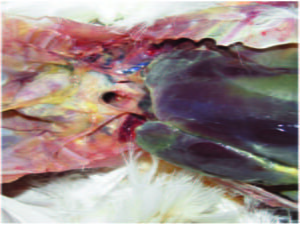
Lesions of Colibacillosis in a Broiler – Caseous pericarditis and perihepatitis
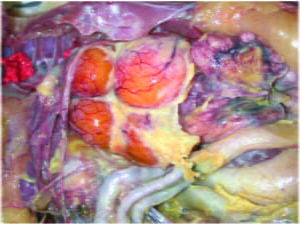
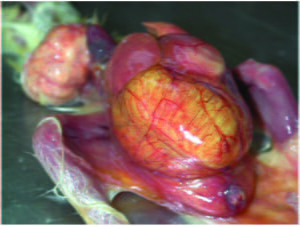
Typical lesions of colibacillosis in layers – Caseous Peritonitis
Prevention and Treatment:
Treatment with antibiotics is normally not used because the course of the disease is so short-lived that by the time a diagnosis is obtained, the disease losses are nearly complete. Effective prevention means only utilizing clean hatch eggs by using effective nest management and sanitation techniques. Also, the use of an effective hatcher and hatcher tray sanitation program is important. The use of Original XPC in feed can also be an aid as E. coli levels in the breeder feces will be less, reducing nest contamination
Treatment now involves supportive measures to aid in reducing the level of infection for the remaining flock members as follows:
- Vaccination with the live E. coli vaccine;
- Fogging (nebulizing) disinfectants such as VirkonS or chlorine dioxide.
- Sanitizing water with five to 10 ppm chlorine or six to 12 ounces of iodine disinfectant per gallon of stock solution metered at one ounce per gallon drinking water.
- Collecting dead birds more frequently to remove them as a source of infection in an attempt to decrease the disease cycle.
- Replacing wet litter with dry in floor houses
- Adding an intestinal health aid such as Diamond V Original XPC will give some immediate boost of the immune system while prolonged use will aid in improving the microflora makeup.
Prevention of E. coli infections is therefore the main control measure used. Several preventative practices are available as follows
• Intestinal health aids. Several research-backed products are on the market. Original XPC has been shown to reduce E. coli levels in the cecum when fed at recommended levels and would aid in reducing contamination levels in the birds’ environment and improve their response to invading bacteria.
• Water sanitation. Routine sanitation of water with either chlorine or iodine can be very effective in reducing bacterial infections.
• Vaccination. The live E. coli vaccine has been a very useful tool in egg layers and is being evaluated in broiler and turkey flocks. In egg layers, two to three doses are applied by spray during the growing period prior to housing. Booster applications are given to some flocks prior to molting.3
• Ventilation practices. Reducing ammonia levels by providing the proper ventilation rates and temperatures throughout grow for broilers will keep litter dry and reduce the release of ammonia from the litter. Keeping the stored manure dry in high-rise layer houses is also important in reducing ammonia. The use of manure drying fans in the pits of highrise houses has been a great aid in reducing moisture of the manure piles.
• Moving age of egg layers. Moving egg layer pullets at least one to two weeks prior to the onset of production will aid in getting the birds’ microflora stabilized and the birds exposed to pathogens in the new layer house environment prior to the stress of egg production. Spreading out these stressors appears to reduce the problems with early lay E. coli problems in egg layers.
References:
Altekruse, S. F., Elvinger, F., Lee, K. Y., Tollefson, L. K., Pierson, E. W., Eifert, J. and Sriranganathan, N. 2002. Antimicrobial susceptibilities of E. coli strains from a turkey operation. Journal of American Veterinary Medicine Association, 221: 411-416.
Barnes, H. J., Vaillancourt, J. P. and Gross, W. B. 2003. Colibacillosis. In: Diseases of Poultry, Y. M. Saif Editor in chief , 11th edition. Iowa State University Press, Ames, IA, USA. Pp 631-652.
Bettelheim, K. A. 1994. Biochemical characteristics of Escherichia coli. In C. I. Gyles Ed., Domestic Animals and Humans pp. 3-29. Wallingford: CABI Publishing.
Harpeet, S. B., Dash, B. B., Sanjeev, K., Singh, H. and Kumar, T. 1993. Motility pattern in indigenous guinea fowl under confinement rearing. Indian Journal of Poultry Science, 28: 63-70.
Hines, M. E., Styler, E. L., Baldwin, C. A. and Cole, J. R. 1995. Combined adenovirus and rotavirus enteritis with E. coli septicaemia in an emu chick. Avian Diseases, 39: 646-651.
Donnenberg, M. S. 2005. Enterobacteriacae. In: Mandenn, G. L. Bennett, J. E.; Dolin, R.; Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases, 6th Edition. Elsevier Churchill Livingstone, Philadelphia. 2: 25672586.
McKissick, J. C. 2006. Poultry Industry Outlook. The University of Georgia, Athens, USA. Available:http://www.ces.uga. edu/Agriculture/agecon/outlook/poultry/ 2006% Accessed October 27, 2006.
Oyetunde, O. O. F., Thomson R. G. and Carlson, H. C. 1978. Aerosol exposure of ammonia, dust and Escherichia coli in broiler chickens. Canadian Veterinary Journal, 19: 187-193
Swarbrick, O. 1985. Pheasant rearing: Associated husbandry and diseases problem. Veterinary Research, 116: 610617.
Welish, R. D., Neiman, R. W., Vanhoosar, S. L. and Dye, L. B. 1997. Bacterial infection of Ratites. Veterinary Medicine, 92: 992-998.
Whiteman, C.E., Bickford, A.A. & Barnes, H.J. (1989). Avian Disease Manual , 3rd edn. Dubuque, IA: Kendall/Hunt.
Yogaratnum, V.1995. Analysis of the causes of high rate of carcass rejection at poultry processing plant. Veterinary Research, 79: 810-816.
Zhou, B., L. I, Y., Wu, T., Hou, B. T. and Wu, T. 1995. Colibacillosis in quails. Chinese Journal of Veterinary Science Technology, 25:34-35.


