HOW TO PREVENT COCCIDIOSIS IN

Sachin Patidar1 Neeraj Kumar*2, Jayshree Jakhar3 and M.K. Verma4
1Research Scholar, Department of Veterinary Parasitology,2,3Research Scholar, Department of Veterinary Pathology,4Ph.D. Scholar, Department of Veterinary Pharmacology and Toxicology,GB Pant University of Agriculture and Technology, Pantnagar, (Uttarakhand)
INTRODUCTION
Poultry coccidiosis is one of the most prevalent serious diseases affecting poultry production worldwide. Poultry producers all over the world invest large resources in coccidiosis prevention and treatment, which results in huge economic losses owing to malabsorption, poor feed conversion rate, lower weight growth, and higher mortality. Coccidiosis is one of the most economically devastating infectious diseases of deep litter-raised chicken and one of the most difficult to control. Parasitic organism infects and damages the digestive tract of the host, resulting in reduced production. It primarily affects the caecal and gastrointestinal systems and is associated with severe morbidity and mortality. Poultry coccidia are often host specific, with different species affecting different parts of the intestine in different ways. All age groups are vulnerable to this disease. Older birds act as carriers of infection, contaminating the environment and become a source of infection consequently.
ETIOLOGY
The coccidia are a diverse group of single-celled parasitic organisms belonging to the sub-kingdom Protozoa that are found around the world. The coccidia of the genus Eimeria are predominantly host-specific, meaning that each species only occurs in one type of host. They show a wide variation in their pathogenicity. Common Eimeria species including
• E.tenella (Caecal coccidiosis)- Most pathogenic, very common & cause caecal coccidiosis.
• E.brunetti (Rectal coccidiosis)- Highly pathogenic, less common & cause rectal coccidiosis.
• E.necatrix (Intestinal coccidiosis)-Highly pathogenic, very common & cause intestinal coccidiosis
LIFE CYCLE AND TRANSMISSION
Coccidiosis is widely found in chicken-raising areas. Coccidia can proliferate quickly inside the intestine or caeca cell lining. Many species of the genus Eimeria may infect chickens with no cross-immunity. Eimeria are tissue and host specific, with a self-limiting life cycle. As observed in wild birds, insects, and rodents, they mechanically transmit sporulated oocysts through contaminated footwear, clothes, equipment, or dust. The most common way for an infection to spread is by direct oral transfer. The life cycle is brief and begins with sporulated oocysts. Following ingestion of sporulated oocysts, the mechanical and acidic environments of the host digestive system induce oocyst excystation in the gizzard, culminating in the release of sporozoites and sporozoites that penetrate and kill cells in the intestinal mucosa and initiate the reproductive cell cycle.
SUSEPTABILITY
It is a serious disease of domestic fowl, turkeys, and ducks. All age groups are susceptible to coccidiosis. Chickens between the ages of 4 to 8 weeks are the most susceptible to infection. Because the parasite has a lifespan of 4-7 days, young birds are more vulnerable to infection than older birds. Coccidiosis rarely occurs in layers and breeders as they have generally acquired immunity to this disease. The disease is ranked just after Salmonella pullorum as it causes huge mortality. The severity of an infection is determined by the age of the birds, the amount of sporulated oocysts consumed, the flock’s immunological state, and environmental management.
CLINICAL SIGN
Infected birds typically exhibit a typical ‘sick bird’ attitude, such as despondency, prostration, huddling together, and ruffled feathers. Common clinical findings include a decrease in feed and water intake, a decrease in growth rate, weight loss, egg production, severe bloody diarrhea, dirty vents, and watery to yellowish or red droppings. Mild infections (subclinical) may cause depigmentation and potentially lead to secondary infection. Surviving birds of severe infections recover in 10–14 days but never recover lost performance.
POST-MORTEM LESIONS
Post-mortem lesion of coccidiosis depends on type of Eimeria species. E. Acervulina lesions show as white patches or lines inside the intestine visible from the outside. The white spots are diagnostic for E. necatrix. Clumps of large schizonts can be demonstrated microscopically. In severe cases, the intestinal wall is thickened, and the infected area dilated to 2–2.5 times the normal diameter. The lumen may be filled with blood, mucus, and fluid. E. maxima lesions are distinguished by multiple petechial haemorrhages, segmental ballooning, or enlargement of the mid-gut with orange-tinted mucus. In E. brunetti infections, coagulative necrosis and sloughing of the mucosa occurs throughout most of the small intestine usually seen. E. tenella lesions are characteristics by hemorrhages surrounding the caecal wall, a thickening of the caecal wall, and a large core of cellular debris and blood.
DIAGNOSIS
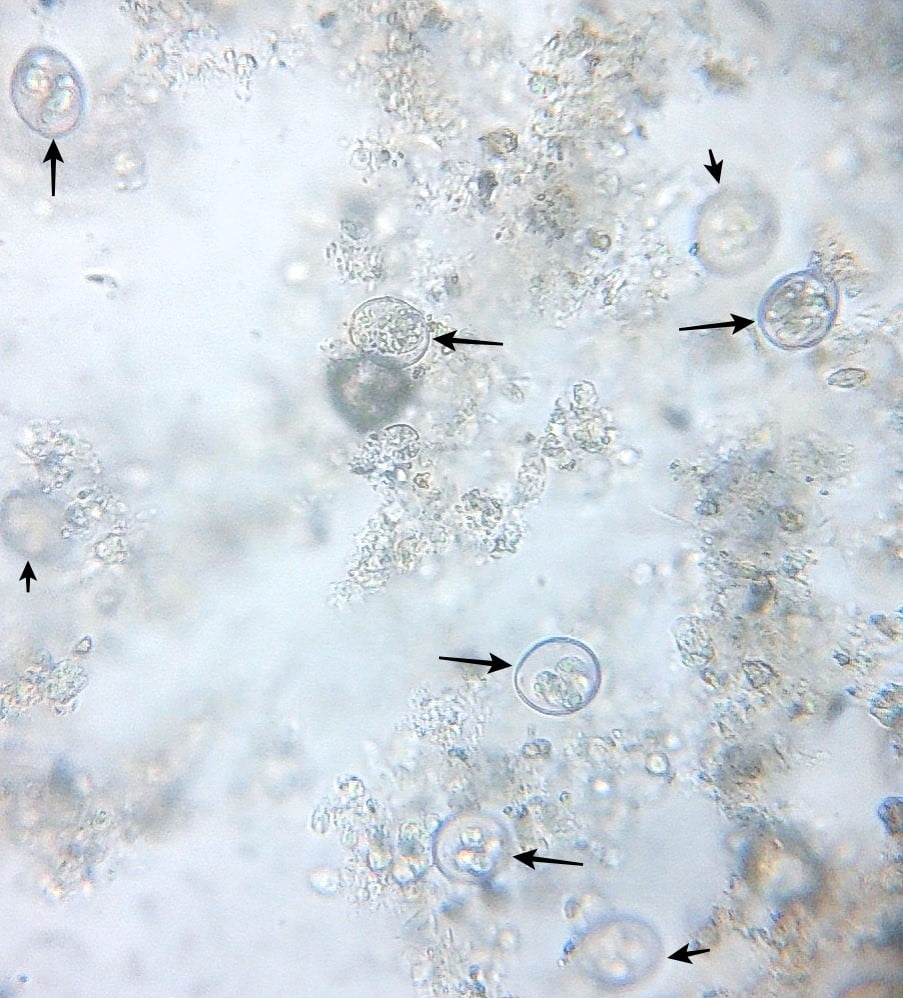
Coccidiosis can be diagnosed by history of infected birds, clinical manifestations, gross lesions during port-mortem and microscopic examination of faecal sample. PM lesions are present along the intestinal tract and often have a distinctive location and appearance that is useful in diagnosis. The location in the host, appearance of lesions, and the size of oocysts are used in determining the species present. Coccidial infections are readily confirmed by demonstration of oocysts in faeces or intestinal scrapings; however, the number of oocysts present has little relationship to the extent of clinical disease. Subclinical coccidial infections may be unimportant, and poor performance may be caused by other flock disorders. Eimeria oocysts are spherical to subspherical, pear-shape, smooth, yellow to light brown in color (Fig.1).
PREVENTION AND CONTROL
• Coccidiosis prevention and control is based on the efficient use of anticoccidial medications or vaccinations, good farm cleaning and disinfection, along with better farm management techniques. In poultry production several antimicrobials or antiprotozoals have been used for decades to treat and prevent coccidiosis (Table 1 & 2).Some examples of anticoccidial antimicrobials including
• Sulfonamides- Sulfadimethoxine, sulfaquinoxaline, or sulfamethazine, but they should not be used in layer hens.
• Ionophores, which influence membrane function of the parasite and act as both coccidiocides and coccidiostats e.g., Monensin
• Quinolones, which influence energy metabolism of the parasite and act as both coccidiocides and coccidiostats e.g., buquinolate, decoquinate, and methylbenzoquate.
• Coccidiostatic thiamine analogs, which influence co-factor synthesis for the parasite e.g., Amprolium. Amprolium has a safety margin of ~8:1 when used at the highest recommended level in feed.
• Diclazuril and toltrazuril are highly effective against a broad spectrum of coccidia. Diclazuril is used mostly for prevention whereas toltrazuril is used primarily for treatment in the water.
Table 1* Drugs for Prevention of Coccidiosis in Poultry
| Drug | Use Level (% in feed) | Withdrawal Time (days) |
| Amprolium | 0.0125–0.025 | 0 |
| Diclazuril | 0.0001 | 0 |
| Monensin sodium | 0.01–0.0121 | 0 |
| Lasalocid sodium | 0.0075–0.0125 | 3 |
| Clopidol or meticlorpindol | 0.0125–0.025 | 0 |
Table 2*- Drugs for Treatment of Coccidiosis in Chickens
| Drug | Feed or Water | Use Level, Treatment Duration | Withdrawal Time (days) |
| Amprolium | Water | 0.012%–0.024%, 3–5 days; 0.006%, 1–2 wk | 0 |
| Chlortetracycline | Feed | 0.022% + 0.8% calcium, not more than 3 wk | 0 |
| Oxytetracycline | Feed | 0.022% + 0.18%–0.55% calcium, not more than 5 days | 3 |
| Sodium sulfachloropyrazine monohydrate | Water | 0.03%, 3 days | 4 |
| Sulfadimethoxine | Water | 0.05%, 6 days | 5 |
| Sulfamethazine (sulfadimidine) | Water | 0.1%, 2 days; 0.05%, 4 days | 10 |
| Toltrazuril | Water | 25 ppm, 2 days | NAb |
*In Aiello, S. E., In Moses, M. A., & In Allen, D. G. (2016). The Merck veterinary manual.
• Anticoccidial properties have been found in certain plant extracts. The effect of 15 different herbs was evaluated, and it was discovered that Ulmus macrocarpa, Pulsatilla koreana, Torilis japonica, Artemisia asiatica, and Sophora flavescens (Kushen) had greater survival rates in day old E. tenella infected broilers than the infected control.
• The use of natural medicines such as fungal extracts, plant extracts, and probiotics to treat coccidiosis has recently improved. Immune stimulation, anti-inflammatory action, antioxidant activity, and cytoplasmic damage are all impacts of several food supplements. Use of nutritional supplement including
* Provide vitamin A: Allows host recovery fast
* Provide Vit. K rich diets: Reduces mortality
* Mix some vital minerals in optimum ratio in ration.
*Provide antioxidants: Antioxidant molecules plays an important role to control and reduce oxidative stress caused by increased levels of reactive oxygen species and free radicals. e.g., Curcumin extracted and vitamin E.
• The use of live vaccines for the control of coccidiosis in broiler breeders or layers is well established. Their use in broilers has been slow to gain acceptance considering the economics, perceived adverse effect on chick growth with virulent vaccines and concerns about timely onset of protective immunity in short lived birds. Commercial vaccines consist of live, sporulated oocysts of the various coccidial species administered at low doses. Modern anticoccidial vaccines should be given to day-old chicks, either at the hatchery or on the farm. Because the vaccine serves only to introduce infection, chickens are reinfected by progeny of the vaccine strain on the farm. Most commercial vaccines contain live oocysts of coccidia that are not attenuated. Commercially available vaccine are 1. Live virulent vaccine- Coccivac and Immucox, 2. Live attenuated Vaccine- Paracox and Livacox.
• Managemental control: Because coccidial oocysts are ubiquitous and easily disseminated in the poultry house environment and have such a large reproduction potential, it is very difficult to keep chickens coccidia free, especially under current intensive rearing conditions. Management has always been important to coccidiosis control, and it focuses on reducing the number of coccidia to keep infection at a minimum until immunity is established. Some good managemental practice objectives including
* Cleaning and fumigation of poultry house.
* Washing of all utensils with chemicals.
* Prefer cage system of poultry rearing.
* Proper disposal of poultry beats.
* Avoid contamination of feed, water and utensils with poultry beats.
* Frequent removal of bedding materials and exposed against direct sunlight.
* Restricted entry of outsiders.
• Depending on the type of poultry production, the approaches for an effective control of coccidiosis are different. Because coccidial oocysts are resistant to environmental conditions and some disinfectants, elimination of coccidiosis by litter cleaning and disinfections is not possible on chicken farms. A straight program is defined by the addition of starter and grower feed. During the straight program, also known as step-up program, the concentration of ionophore in grower feed can be raised for maximal protection at the period of peak coccidian oocyst shedding (3-4 weeks). Step-down programmes relate to the reduction in anticoccidial concentration in grower or finisher feed. Shuttle program refers to the inclusion of chemical and ionophore anticoccidials to starter and growth feeds, respectively.


