ROLE OF PHYTIC ACID AND PHYTASE IN POULTRY NUTRITION
Prakash Sannamani, Jayanaik, K. Venkata Reddy and K. Rengarajan
Department of Poultry Science, Veterinary College, Bangalore – 560024
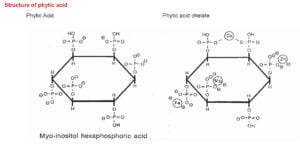
The major feed ingredients that are commonly used in commercial poultry feed are cereal grains, oil seed meal and grain by-products. A major portion (60 – 80%) of the total phosphorus present in these ingredients, occur in the form of phytic acid. Phytic acid (phytate) is a complex of Ca or Mg with myo-inositol (Cosgrove, 1980) and is an essential component of all seeds. Phytic acid is considered as anti nutritional factor in poultry because it binds with phosphorus and other important nutrients and thereby decreases their availability (Cabahug et al., 1999).
Structure of phytic acid
Occurrence of Phytic Acid
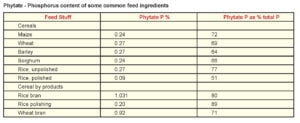
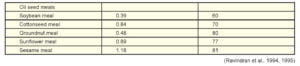
Among common feed stuffs oil seed meals and cereal by-products contain large amount of phytate phosphorus whereas cereal grains and grain legumes contain moderate amounts. Aleurone layer is the major site of phytate deposition in cereals and in dicotyledons seeds phytate is distributed throughout the kernel (Lott, 1984). In rice more than 80% of the phytate is present in the outer bran and in maize around 90% of the phytic acid is concentrated in the germ portion (O’Dell et al., 1972). Among the common feed stuffs, sesame meal and rice bran have the highest level of phytate. Concentrate of phytate in poultry rations ranges from 2.5 to 4 g/kg (Ravindran et al., 1995).
Phytate and Minerals Bio – Availability
About 2/3 of the total phosphorus present in plant feed ingredients is in phytate form (Simons et al., 1990). Each phytic acid molecule contains 28.2% phosphorus. Phosphorus is very low for poultry because they have limited capability to utilize phytate- phosphorus. Capability of the poultry to utilize phytate phosphorus ranges from 0 – 50% (Singh et al., 2003a, b). Bioavailability of P in maize and soybean meal for poultry ranges from 10 – 30%.
Low availability of phytate phosphorus in poultry results in two problems:
- The need to add inorganic phosphorus supplements to diets and
- Excretion of large amounts of phosphorus into the nature which is responsible for environmental pollution.
Phytate forms complexes with cations in the following descending order: Cu2+>Zn2+>Co2+>Mn2+>Fe3+ >Ca2+ (Vohra et al. 1965). One molecule of phytic acid binds with 3 to 6 molecules of Ca to form insoluble complex at the pH of small intestine. Excess Ca in the diet leads to the formation of calcium phytate complexes which are highly insoluble and poorly digested. Zinc forms the most insoluble salt with phytic acid, therefore zinc is a limiting mineral in high phytate diets as it forms a highly insoluble complex at pH 6.4, which is the pH of the upper intestine where most mineral absorption occurs (Maddaiah et al., 1964).
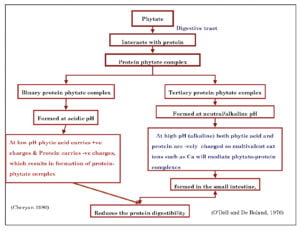
Effect on Protein Bio-availability
Phytate forms complex with the protein and decreases their availability. Protein-phytate complexes are formed in the GIT (Ravindran et al., 2000). The formation of protein- phytate complex begins in the seed during ripening when phytate accumulates primarily in the aleuronic layer of monocotyledonous seeds and in the protein bodies of dicotyledonous seeds (Ravindran et al., 1995).
Phytate Interaction with Digestive Enzymes
Phytate inhibits digestive enzymes such as pepsin, alpha- amylase and trypsin (Deshpande and Cheryan, 1984). Phytate inhibit the proteolysis by altering the protein configuration of digestive enzymes (Singh and Krikorian, 1982). Phytate binds with trypsin via Ca forming a tertiary complex and thereby inhibits the trypsin activity. Inhibition also results from the chelation of Ca ions which are essential for the activity of trypsin and alpha-amylase (Liener, 1989).
Degradation of Phytic Acid
Phytic acid decreases the bioavailability of P, Ca, Mg, Zn and many other trace elements. Hydrolysis of phytic acid occurs through various processing effects or by phytase enzyme. Phytase is a phosphatase enzyme which hydrolyses phytic acid to inositol and inorganic phosphorus, leading to improved phosphorus utilization (Singh et al., 2003).
Phytic acid ____Phytase_> Inositol + Inorganic Phosphorus
Poultry lacks endogenous phytase enzyme, therefore P, Ca protein and other phytic acid bound nutrients are less available to poultry.
Sources and Activity of Phytase
Phytase is widely distributed in plants, animals and microbes (Sebastian et al., 1998). The four sources of phytase are: (1) Intestinal phytase found in digestive secretions. (2) Phytase produced by endogenous microbes in the digestive tract (e.g. ruminants). (3) Phytase produced by exogenous micro-organisms, and (4) Endogenous phytase from plant feed-stuffs. The Nomenclature Committee of International Union of Biochemistry (1979) lists two phytase i.e, 3-phytase and 6- phytase.
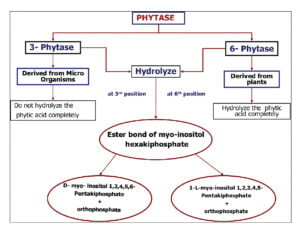
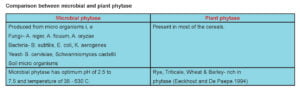
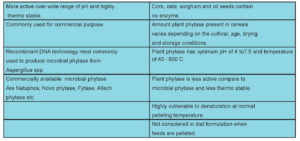
Among all sources Aspergillus niger is the most active source of high phytase activity and Aspergillus ficuum produces highest concentration of phytase and phytase from A. ficuum is highly thermo stable. In mammals, phytase is produced in the brush border region of small intestine but the concentration is very less (Cooper and Gowing.1983). In poultry also phytase is produced from brush border region of intestine but the activity is of little significance (Maenz et al.1997). The activity of phytase is measured in terms of inorganic phosphorus released from phytic acid and the activity is known as phytase unit (FYT). One phytase unit is the amount of enzyme needed to release 1 µ mol of inorganic phosphate/ min from 5.1mM Sodium phytate at pH 5.5 and 37o C (Engelen et al., 1994).
Site of Phytase Activity
Crop and proventriculus are the main sites of phytase activity in poultry. In chickens, 69 to 86 per cent microbial phytase activity is observed in the crop and 31 to 38 % in the proventriculus. No phytase activity was detected in the small intestine.
Nutritional Benefits of Phytase
Phytase increases bioavailability of P, Ca and amino acids. Supplementation of microbial phytase increases body weight gain, feed intake, feed efficiency and overall growth performance in broiler chickens (Singh and Khatta, 2002; Singh et al., 2003, Karim 2006). Nelson et al. (1971) were the first to supplement 0.4% crude phytase (produced by A. ficcum) in corn soy based broiler diets and noticed a 33.3% improvement in BWG. The improvement in growth performance in chickens fed with phytase may be due to increased bioavailability of phosphorus by phytase, increase in feed intake and feed efficiency, increase utilization of inositol (Simons et al., 1990), improvement in starch digestibility (Knuckles and Betschart, 1987), improved utilization of protein and amino acids and overall utilization of nutrients.
Availability of Protein and Amino Acids
Phytase supplementation improves nitrogen retention in broiler chickens and also improves apparent and true ileal digestibility of nitrogen and amino acids. Phytase also increases the utilization of methionine, lysine, valine, isoleucine and total amino acids in broiler diets (Biehl and Baker, 1997). Phytase supplementation improves the protein utilization in poultry by countering the anti-nutritive properties of phytic acid (Selle et al., 2007).
The factors affecting protein utilization to added Phytase: a) Concentration of phytate in feed ingredients: Magnitude of response to phytase is directly proportional to concentration of phytate in ingredients and poorly digestible feedstuffs show more response to phytase than those with higher inherent digestibility (Ravindran et al., 1995). b) Source and concentration of protein in the diet. c) Digestibility of the protein. d) Ca and P levels in the diet, and e) Rate of inclusion phytase.
Energy Utilization
Phytase has a positive effect on energy parameters. Supplementation of phytase increases AME & TME (Selle et al., 2007). Improved energy utilization in poultry diets added with phytase is mainly due to increase in protein/amino acid digestibility.
Effect of Phytase on Phosphorus and Calcium bioavailability
Main aim of using microbial phytase is to release of phosphorus from plant derived feed ingredients. Supplementation phytase increases the phosphorus availability from 20 to 40%, decreases the phosphorus excretion by 50% and also improves serum in organic phosphorus concentration in broilers. The amount of phytate phosphorus released by microbial phytase depends on: a) Concentration of phytate in the diet (Ravindran et al., 1995) b) Concentration and source of the added phytase (Kornegay et al., 1996). Supplementation of phytase @ 500 FYT/Kg to low phosphorus diet increased phosphorus retention by 12.4% similarly supplementation of phytase @ 800 FYT/Kg to wheat-sorghum-soy based diet increased availability of P by 40.3 % (low), 58.9 % (medium) 44.1% (high) phytate phosphorus diet, and c) Ca and Vitamin D3 contents (Qian et al. 1995).
Addition of vitamin D3 along with phytase increases the utilization of phytate (Mitchell and Edwards, 1996). Supplementation of microbial phytase increases the availability of Ca in broiler chicks (Singh et al., 2003). Retention Ca increases as the level of phytase increases in the diet and decreases as the ratio of Ca: P become wider in the diet (Qian et al., 1996). Phytase increases tibia or toe ash in broilers, this may be due to increased bioavailability of phosphorus and Ca by phytase (Nelson et al., 1971).
Availability of Other Minerals
Phytic acid has the strong chelating potential to form complexes with cations and their by decreases their availability. Phytase hydrolyzes the phytic acid and releases variable amount of bound minerals. Supplementation of microbial phytase increases the retention of Zn from 27.6% to 34.7%. Phytase improves Zn utilization by reducing Zn excretion in broilers and there by reduces requirement of Zn in broiler diet (Jondreville et al., 2007). Phytase increases the retention of Cu from 2.46% to 5.4% in broilers fed low P diet and improves utilization of Mn in broiler chicks (Mohanna and Nys.1999).
Performance of layers
In layers supplementation of phytase to low phosphorus diet is very effective as a replacement for in organic phosphorus. Phytase supplementation in low phosphorus diet improves bioavailability of phytate phosphorus resulting an increase in feed intake, egg production, egg weight and egg shell thickness.
Economics of Phytase Supplementation
Phosphorus is one of the costliest item in poultry feed. Replacement of inorganic phosphorus by phytase enzyme leads to more economic poultry production. Supplementation of phytase @ 25 g /100 kg in corn- soy-wheat bran based diets without DCP reduced the feed cost (Vinil et al., 2000). Supplementation of broiler diets with phytase @ 300 gm / ton of feed without DCP resulted in increase in net income by 9.47% (Kundu et al., 2000).
Conclusions
- Phytate phosphorus utilization by poultry varies from 0 – 50% depending upon the source of phytate, age of birds, and dietary levels of Ca and vitamin D3.
- Due to its strong chelating potential, phytic acid binds with essential minerals (Ca, Zn and Cu) and protein makes them unavailable for absorption.
- Phytase has the capacity to counteract the anti – nutritional effects of phytate by hydrolyzing the phytic acid to inositol and inorganic phosphorus.
- Supplementation of microbial phytase along with feed ingredients improves the growth performance and retention of P, Ca, & Cu when fed to poultry.
- Phytase also increase the AME and ileal digestibility of protein and amino acids. Therefore supplemental microbial phytase can be used in poultry ration to increase the growth performance, digestibility of nutrients and to reduce the feed cost.
REFERENCES
BIEHL, R.R. AND BAKER, D.H. (1997) Microbial phytase improves amino acid utilization in young chicks fed diets based on soyabean meal but not diets based on peanut meal. Poultry Science 76: 355-360.
CABAHUG, S., RAVINDRAN, V., BRYDEN, W.L. AND SELLE, P.H. (1999) Response of broilers to microbial phytase supplementation as influenced by dietary phytic acid and non-phytate phosphorus levels. I. Effects on broiler performance and toe ash content. British Poultry Science 40: 660-666.
CHERYAN, M. (1980). Phytic acid interactions in food systems. CRC Critical Reviews in Food Science and Nutrition 13: 297-302.
COOPER, J.R. AND GOWING, H.S. (1983) Mammalian small intestine phytase (EC 3.1.3.8). British Journal of Nutrition 50: 673-678.
COSGROVE, D.J. (1980) Inositol Phosphates: Their Chemistry, Biochemistry and Physiology. Elsevier Publication Scientific Publishing Co., New York.
DESHPANDE, S.S. AND CHERYAN, M. (1984) Effects of phytic acid divalent cations, and their interactions on alpha-amylase activity. Journal of Food Science 49: 516-519.
EECKHOUT, W. AND DE PAEPE, M. (1994) Total phosphorus phytate-phosphorus and phytase activity in plant feed stuffs. Animal Feed Science and Technology 47: 19-29.
ENGELEN, A.J., VAN DER HEEFf, E.C., RANDSDORP, P.H.G. AND SMITH, E.L.C. (1994) Simple and Rapid determination of phytase activity. Journal of the Association of Official Analytical Chemists International 77: 760-764.
JONDREVILLE, e., LESCOAT, P., MAGNIN, M., FEUERSTEIN, D., GRUENBERG, B. AND NYS, Y. (2007) Sparing erect of microbial phytase on zinc supplementation in maize-soyabean meal diets for chickens. Animal 1(6): 804-811.
KARIM, A. (2006) Responses of broiler chicks to non-phytate phosphorus levels and phytase supplementation. International Journal of Poultry Science 5 (3): 251-254.
KNUCKLES, B.E. AND BETSCHART, A.A. (1987) Effect of phytate and other myo-inositol phosphate esters on alpha-amylase digestion of starch. Journal of Food Science 52: 719-721,
KUNDU, B., BISWAS, P., RAJENDRAN, D. AND DANDOPATH, S.K. (2000) Effect of supplementation of phytase enzyme and gradual replacement of dicalcium phosphate on the performance of commercial broker chicken. In: Proceedings of III Biennial conference of Animal Nutrition Association. Hisar, India. Abst. No. 128.
LIENER, I.E. (1989) Anti nutritional factors in legume seeds: State of art. In: HUISMEN,J.,VAN DER POEL, T.F.B. & LlENER, I.E. (Eds) Recent Advances of Research on Anti- nutritional Factors in Legume Seeds, pp. 6-13 (Pudoc, Wageningen, The Netherlands).
LOTT, J.N.A. (1984) Accumulation of seed reserves of phosphorus and other minerals. In: MURRAY, D.R. (Ed) Seed Physiology. VoU, pp. 139-166 (Academic Press, London).
MADDAIAH, V.T., KUMICK, A.A., HULLET, B.J. AND REID, B.L. (1964) Nature of intestinal phytase activity. Proceedings of the Society of Experimental Biology and Medicine 115: 1054-1057.
MAENZ, D.D., ENGELE-SCHAAN C.M. AND CLASSEN, H.L. (1997) Endogenous phytase activity in the small intestinal brush border membrane of broiler chicks and laying hens. Poultry Science 76 (suppl.): 71.
MITCHELL, R.D. AND EDWARDS, H.M. (1996) Effect of phytase and I, 25-dihydroxycholecalciferol on phytate utilization and the quantitative requirement for calcium and phosphorus in young broiler chickens. Poultry Science 75: 95-110.
MOHANNA, C. AND NYS, N. (1999) Changes in zinc and manganese availability in broilers chicks induced by vegetal and microbial phytase. Animal Feed Science and Technology 77: 241-253.
NELSON, T.S., SHIEH, T.R., WODZINSKI, RJ. AND WARE, J.H. (1971) Effect of supplemental phytase on the utilization of phytate phosphorus by chicks. Journal of Nutrition 101: 1289-1294.
O’DELL, B.L., DE BOLAND, A.R AND KOIRTYOHANN, S.R. (1972) Distribution of phytate and nutritionally important elements among the morphological components of cereal grains. Journal of Agricultural Food Chemistry 20: 718-721.
RAVINDRAN, V., BRYDEN, W.L AND KORNEGAY, E.T (1995) Phytates: Occurrence, bioavailability and implications in poultry nutrition. Poultry and Avian Biology Review 6(2):125 – 143.
SELLE, P.H., RAVINDRAN, V., RAVINDRAN, G. AND BRYDEN, W.L. (2007) Effect of dietary lysine and microbial phytase on growth performance and nutrient utilization of broiler chickens. Asian-Australasian Journal of Animal Sciences 20 (7): 1100-1107.
SIMONS, P.C.M., VERSTEEGH, H.A.J., JONGBLOED, A. W., KEMME, P.A., SLUMP, P., BOS, K.D., WOLTERS, M.G.E., BEUDEKER, R.F. AND VERSCHOOOR, G.J. (1990) Improvement of phosphorus availability by microbial phytase in broilers and pigs. British Journal of Nutrition 64: 525-540.
SINGH, M. AND KRIKORIAN, A.D. (1982) Inhibition of trypsin activity in vitro by phytate. Journal of Agriculture and Food Chemistry 30: 799-800.
SINGH, P.K. and KHATTA, V.K. (2002) Phytase supplementation for economic and eco-friendly broiler production. Journal of Eco Physiology 5 (3-4): 117-121.
SINGH, P.K., KHATTA, V.K. AND THAKUR, R.S. (2003) Effect of phytase supplementation in maize based diet on growth performance and nutrients utilization of broiler chickens. Indian Journal of Animal Sciences 73 (4): 455-458.
VINIL, S.P., KADIRVELAN, C., THIRUMALAI, S., VALLI, S. AND CHANDRABOSE, B. (2000). The role of phytase and other enzymes in augmenting the bioavailability of nutrients in broilers. Proceedings of XX Annual conference of Indian Poultry Science Association, Chennai, India. Abst. No. PPN. 97.
VOHRA, P., GRAY, G.A. AND KRATZER, F.H. (1965). Phytic acid-metal complexes. Proceedings of the Society of Experimental Biology and Medicine 120: 447.





This thepoultrypunch.com and ROLE OF PHYTIC ACID AND PHYTASE IN POULTRY NUTRITION article has helped
me a lot, is very well written, I’m glad I found it. My friend Shanizrrerc and
I used best weight loss product: https://s96.me/fit and we reached the ideal weight.
I’m so happy now! 🙂 Kiss you all!